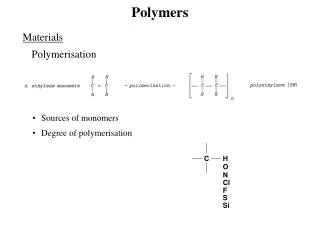
Polymers
Polymers. Polymers. Large chain like molecules that are built from small molecules called monomers . Many important biomolecules are also natural polymers. (know these!) Protein (made up of amino acids ) DNA (made up of nuclei acids ) Starch (made up of sugar subunits)


Polymers
E N D
Presentation Transcript
Polymers • Large chain like molecules that are built from small molecules called monomers. • Many important biomolecules are also natural polymers. (know these!) • Protein (made up of amino acids) • DNA (made up of nuclei acids) • Starch (made up of sugar subunits) • cotton, silk and cellulose.
Polyethylene • The simplest and best known synthetic polymer is polyethylene. • Ethylene polyethylene (Monomer) (polymer)
The properties of a polymer can be varied by substituting other elements or groups of elements for the hydrogens on ethyelene. • Example - tetrafluoroethylene has fluorine atoms instead of Hydrogen atoms and is used to make Teflon which is used in nonstick coatings for cooking utensils
Polymers have different properties based on the side chain attachments, because these side chains will have different strengths of intermolecular attractions depending on their polarity. • The attractions between these long molecules determines the polymers flexibility, hardness or softness and melting temperature.
SLIME! • A Cross-linked polymer • Made from Polyvinyl Alcohol • Crosslinked with sodium borate(Borax) • Creates intermolecular bonds between polymer chains.
Cross linked Polyvinyl Alcohol • SLIME!!!!