polymers
polymers. Dr. Waseem Bahjat Mushtaha Specialized in prosthodontics. Definitions:. a) polymers: are long chain molecules consisting of many repeating units. b) A monomer: is the smallest repeating unit in the polymer chain.


polymers
E N D
Presentation Transcript
polymers Dr. Waseem Bahjat Mushtaha Specialized in prosthodontics
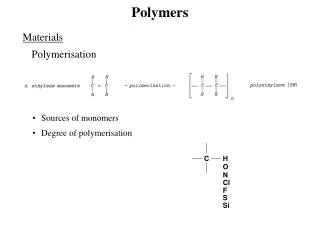
Definitions: a) polymers: are long chain molecules consisting of many repeating units. b) A monomer: is the smallest repeating unit in the polymer chain. c) polymerization: is the chemical reaction by witch polymers are formed from monomers.
Types of polymers: I- According to the origin: a) Natural polymers e.g., natural rubbers, polysaccharide, DNA, silk, agar, alginate. b) Synthetic polymers i.e. produced in the laboratory by chemical reactions .e.g. acrylic resin, nylon
II- According to the thermal behavior: a) Thermoplastic polymers: i.e. soften when heated and harden when cooled (reversable or physical reaction) heating Hard soft cooling e.g. polymethyl methacrylate.
ii- thermosetting polymers: i.e. harden during fabrication (irreversable or chemical reaction. chemical Soft Hard reaction e.g. Cross-linked poly methyl methacrylate.
iii- According to the spatial structure: In addition to the chemical composition, the physical or spatial structure of the polymer is very important in determining the properties of the polymer. There are three basic types of structures:
a) Linear polymers: this may be homopolymer (simple polymer: i.e. has one type of monomer, or copolymer, this means that it has more than one type of monomer. Linear copolymer may be random or block. b) Branched polymers: this type of polymers show higher strength, hardness and less water sorption than linear polymer. Branched polymers are either homo or copolymer. The branched copolymer may be random or graft types c) Cross- linked polymers.
III- According to the polymerization reaction: Polymerization: the chemical reaction by witch polymers are prepared from monomers. polymerization Monomer polymer reaction
Types: 1) Condensation polymerization: The reaction between two molecules to form a larger molecule with the elimination of a smaller molecule such as water as a by – product. condensation monomer polymer+ by product polymerization
An example for condensation polymerization is a phenol formaldehyde polymer known as ‘Bakelite’ which has used in dentistry in the past as denture base. Another example of a condensation polymerization reaction used in dentistry is the polysulphide rubber impression material.
2) Additional polymerization: The reaction between two molecules to give a larger molecule without the elimination of a smaller molecule ( no by- product). There is no change in the composition, the structure of the monomer is only repeated many times. addition Monomer polymer polymerization
Stages of addition polymerization: The addition polymerization reaction passes through 3 stages: 1- Activation and initiation 2- Propagation 3- Termination
1- Activation and initiator: The addition polymerization reaction requires the presence of free radicals. A free radical is very reactive compound with an unpaired electron. Before initiation occurs, the initiator (benzoyl peroxide) has to activated . Activation of the initiator is done by light (ultraviolt or visible blue light), heat, or by a chemical compound such as dimethyle para - toluidine. In this way the benzoyl peroxide initiator decomposes giving up free way the benzoyl peroxide initiator decomposes giving up free radicals to initiate the polymerization reaction. Activation R-R 2R “Free radicals” Heat or chemical or light .
.. 2- propagation: R + M “stable monomer” RM “Active monomer The chain reactions continue with the evolution of heat, until all the monomer is changed to polymer. RM + M RMM+M RMMM and so on until RMn where n is integral number.
3- Termination: The chain reaction can be terminated either by: a) Direct coupling: This occurs when two free radicals react to form a stable molecule: R Mn M +R R MN+1 R
B- Exchange of hydrogen atom: from one growing chain to another H H H H R Mn C C R Mn C C H H H H H + + H H H H R Mm C C R Mm C C H H H
Factors associated with polymerization: Polymerization reactions are associated with: a) Evolution of heat as the reaction is strongly exothermic. b) Reduction in volume. This is about 21% of monomer, but for a polymer monomer mixture it will decrease to 6 to 7%. All this methods of manipulation of acrylic resin attempt to overcome this problem of heat and shrinkage.
Inhibition of polymerization: Any impurity in the monomer which can react with free radicals will inhibit or retard polymerization reaction. it can react either with the activated initiator or any activated nucleus, or with an activated growing chain to prevent further growth . The presence of such inhibitors influences the length of the initiation period, as well as the degree of polymerization.
For example, the addition of a small amount of hydroquinone to the monomer will inhibit polymerization if no chemical initiator is present, and it will retard the polymerization in the presence of an initiator. Also oxygen and zinc-oxide eugenol can inhibit polymerization
Degree of polymerization and molecular weight The degree of polymerization may be defined as the number of repeating units in a polymer, i.e. the number of monomer units joined together. The molecular weight is equal to the degree of polymerization multiplied by the molecular weight of the repeating units.
It should be noted that the growth of polymer chains is a random process, some chain grow faster than others( some are terminated before others. Thus not all chains have the length ) they have a distribution of molecular weights. The quoted molecular weight for a polymer is an average value.
Copolymerization: To modify the properties of polymer, it is often advantageous to use two or more chemically different monomers, such a polymer is called a copolymer. Co-polymerization processes enabled chemists to “tailor – make” molecules of predicted properties for special applications.
Examples : a) Addition of sufficient amounts of octyl methacrylate results in a copolymer that is soft and flexible at mouth temperatures and that has been used as a soft liner which will be described later. b) Hydroxy -ethyl methacrylate, increase the water sorption and wettability of the copolymer by saliva. The material becomes soft and flexible if placed in water or saliva; these materials have found applications as soft contact lenses and as soft liners for dentures.
Physical state of polymers: Dental polymers exist at room temperature either as rubbers (elastomers) or hard. Rubber consist of long chain molecules that coiled. When the material is stretched the only work done is uncoiling of the molecules. Thus such materials are easy to deform. This deformation is largely reversible.
Although rubbers are rubbery at room temperature, the effect of intermolecular forces will increase as temperature decreases, and at a certain temperature (the glass transition temperature Tg ) the intermolecular forces become so large as to inhibit uncoiling . Thus below the glass transition temperature the material will be rigid. Likewise if a hard polymer is heated, it looses rigidity at a certain temperature and becomes rubbery
Cross linking: It is chemical bond formed between polymer chains. A chemical compound with two double bonds per molecule can act as a cross linking agent, since each C = C bond can react with a different chain. This produces superior properties. An example of a cross linking agent is ethylene glycol dimethacrylate.
The effect of cross-linking: A small degree of cross-linking limits the amount of movement of the polymer chains relative to each other when the material is stressed . Thus (a) the deformation is elastic rather than plastic (b) The polymer may have a higher value of glass transition temperature. Extensively cross-linked polymers are (c) harder ,more brittle and (d) more resistant to the action of solvents , to surface cracking ,or crazing , in the mouth than non-cross-linked.
Plasticisers: Liquids are able to penetrate between the chain of polymer, the molecules become further apart, and the forces between them become less. Such liquids are called plasticisers. They (a) soften the material and (b) make it more flexible by lowering its glass transition temperature.
Plasticizers • Bleaching Tray
Uses of polymers in dentistry 1) Denture base materials 2) Acrylic teeth for partial and complete dentures. 3) Impression materials: agar, alginates and rubbers. 4) Soft linings 5) Pit and fissure- sealants 6) Composite filling materials.
7) Cements (zinc polycarboxylate and glass ionomer). 8) Varnishes. 9) Temporary crown and bridge materials. 10) Mouth protectors. 11) Root canal filling materials.
Rubbers • putty rubber soft rubber