Understanding Cellular Respiration: Photosynthesis, Redox Reactions, and Biochemistry Basics
This presentation provides an in-depth exploration of cellular respiration, highlighting its relationship with photosynthesis, redox reactions, and the biochemical foundations of life. It discusses how photosynthesis converts CO2 and water into glucose using sunlight, whereas respiration involves the oxidation of glucose back into CO2, releasing energy. The presentation reviews essential macromolecules, enzymes, and catalysis that play critical roles in these processes. By understanding these concepts, we can appreciate the interconnectedness of life functions and energy transformations.

Understanding Cellular Respiration: Photosynthesis, Redox Reactions, and Biochemistry Basics
E N D
Presentation Transcript
Cellular Respiration LA Charter School Science Partnership 28 Apr 2012 Nick Klein
Today’s Talk • Part 1: Big picture: review of photosynthesis, redox • Part 2: Macromolecules, enzymes, and catalysis • Part 3: Respiration & Fermentation
Part 1: The big picture • Let’s think back to photosynthesis. • Photosynthesis is the process by which organisms use the energy in sunlight to chemically transform carbon dioxide (CO2) into organic carbon compounds such as sugars 12H2O + 6CO2 C6H12O6 + 6O2 + 6H2O
Part 1: The big picture • Photosynthesis and respiration both involve reduction/oxidation (redox) reactions— chemical reactions that involve the movement of electrons from one molecule to another • In photosynthesis, when carbon dioxide is fixed, it is reduced (electrons are added to it) which produces organic carbon compounds
Part 1: The big picture Loss ofElectrons isOxidation goesGain ofElectrons isReduction
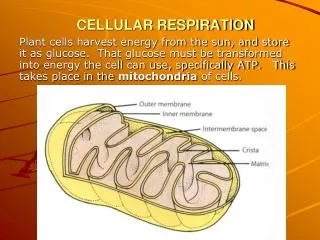
Part 1: The big picture • Respiration is in many ways photosynthesis BACKWARDS. Photosynthesis uses sun energy to turn CO2 into glucose. Respiration releases that stored energy from glucose. C6H12O6+ 6O26CO2+ 6H2O
Part 1: The big picture • So if photosynthesis involves the chemical reduction of CO2 into glucose, and respiration is very similar to photosynthesis backwards… • Respiration is the oxidation of glucose back into CO2, which releases the stored chemical energy!
Part 1: The big picture • Organisms that make their own food are called autotrophs. Organisms that make food using photosynthesis are photoautotrophs • All animals, including humans, are heterotrophs—we have to consume other organisms as food
Part 1: The big picture • Photosynthesis respiration work together in what is called the carbon cycle
Part 1: The big picture Image courtesy NASA Earth Observatory
Part 2: Macromolecules & Catalysis • Before we get to the details of cellular respiration, let’s cover a few more basics of biochemistry that will help us understand both photosynthesis and respiration better! • Specifically, we’re going to briefly discuss the basic building blocks and machinery of biology
Part 2: Macromolecules & Catalysis • What are the basic building blocks of life? • Amino acids (proteins) • Sugars (carbohydrates) • Lipids (fats) • Nucleic acids (DNA & RNA) • All of these “building blocks” string together to form chains called macromolecules or biopolymers
Part 2: Macromolecules & Catalysis • Remember glucose? Glucose is the basic unit of a large number of different sugars (carbohydrates)
Part 2: Macromolecules & Catalysis • Glucose can bond with other glucose molecules in several different ways Sucrose (table sugar)
Part 2: Macromolecules & Catalysis • Glucose can bond with other glucose molecules in several different ways Lactose (milk sugar)
Part 2: Macromolecules & Catalysis • Glucose can also form long chains Cellulose (woody part of plants)
Part 2: Macromolecules & Catalysis • Glucose can also form long chains Starch
Part 2: Macromolecules & Catalysis • Amino acids chain together to form proteins Catalase
Part 2: Macromolecules & Catalysis • Nucleic acids chain together to form DNA & RNA DNA
Part 2: Macromolecules & Catalysis • Our body has to break down sugar polymers into the individual sugar monomers (glucose) before we can use it in cellular respiration • Can our bodies use cellulose? Why or why not? • We don’t have the right biochemical machinery to digest cellulose! We would need a cellulase enzyme…
Part 2: Macromolecules & Catalysis • Enzymes are proteins (chains of amino acids) that act as biological catalysts: they speed the rate of a chemical reaction, but are left unchanged by the reaction • Example demo: catalase
Part 2: Macromolecules & Catalysis • Catalase speeds the reaction:2H2O2 2H2O + O2 • What do you think will happen when I pour H2O2 on the potato? Catalase
Part 2: Macromolecules & Catalysis • Enzymes work by lowering the activation energy. The activation energy is a measure of how much chemical energy a molecule must have before it will undergo a reaction. • Enzymes lower this “hill” and cause reactions to happen that would otherwise only go very slowly
Part 2: Macromolecules & Catalysis • Other examples of enzymes: lactase, cellulase, amylase • If you’re lactose intolerant, your body does not produce enough lactase to digest lactose sugar very well • Similarly, we cannot digest the woody part of plants since our bodies don’t produce cellulase—cows and other herbivores have bacteria in their guts that make cellulase
Part 2: Macromolecules & Catalysis • We explored the action of amylase in one of our morning activities
Part 3: Respiration & Fermentation 12H2O + 6CO2 C6H12O6 + 6O2 + 6H2O C6H12O6 + 6O2 6H2O + 6CO2 Respiration is photosynthesis backwards!
Part 3: Respiration & Fermentation Pigments Photosystem 2H2O 4e- + 4H+ + O2
Part 3: Respiration & Fermentation Electron TransportChain Pigments Photosystem 4e- NADPH
Part 3: Respiration & Fermentation ATP + NADPH CO2 C6H12O6 The Calvin Cycle (light independent reactions)
Part 3: Respiration & Fermentation • In photosynthesis, we used light energy to split electrons out of a water molecule, then used the electron transport chain to take energy from those electrons and convert it into ATP • Then we used ATP and the leftover electrons (in the form of NADPH) to fix (reduce) CO2 into glucose using the Calvin Cycle
Part 3: Respiration & Fermentation • In respiration, we oxidize glucose (add oxygen to transform it into 6CO2) to “pull” electrons out of it • These electrons are then put through an electron transport chain to generate ATP • What do we need for respiration? • Glucose • Oxygen
Part 3: Respiration & Fermentation • First step in respiration is glycolysis • In glycolysis, glucose (6 carbons) is split into two molecules of pyruvate (3 carbons each) • This yields 2 ATP and 2 NADH (electron carriers) • If no O2 is available, glycolysis is the only way to get energy from glucose and fermentation occurs
Part 3: Respiration & Fermentation • In fermentation, we get 2 ATP from glycolysis but can’t continue to the Krebs cycle, which requires O2 to function • Have to recycle the NADH, so the electrons the NADH carries are transferred to pyruvate and glycolysis can continue • Different organisms transform pyruvate to different waste molecules in fermentation—in humans, lactic acid.
Part 3: Respiration & Fermentation • But, if we have O2 we can put the pyruvate into the Krebs cycle and yield 38 ATP total instead of 2 for each glucose! • Krebs cycle is complex, but in basic terms pyruvate is added to a 4-carbon molecule to make citrate, which is then oxidized one CO2 at a time • Each time a carbon is removed from citrate, CO2 is produced and we pull electrons out and transfer them to NADH or FADH2
Part 3: Respiration & Fermentation • In the Krebs Cycle, we’ve oxidized pyruvate into CO2 and produced NADH and FADH2 (electron carriers) • These electron carriers now move the electrons to the electron transport chain (remember from photosynthesis?) • As the electrons flow through the transport chain, their energy is used to create a proton gradient • Then, when the protons flow back in, they drive ATP synthase (an enzyme!) which makes ATP
Part 3: Respiration & Fermentation • Recap: in glycolysis, we split 6-carbon glucose into two 3-carbon pyruvate and yield 2 ATP • Stop at glycolysis if no oxygen available, then fermentation • If oxygen is available, Krebs Cycle oxidizes pyruvate and strips the electrons from it • NADH and FADH2 carry electrons stripped from glucose to electron transport chain where they are used to make ATP (energy)