Balancing Equations
Balancing Equations. H 2 + Cl 2 → HCl. Balancing Equations. KI + Br 2 → KBr + I 2. Balancing Equations. CH 4 + O 2 → CO 2 + H 2 O. Balancing Equations. V 2 O 5 + Ca → V + CaO. Balancing Equations. NaCl + F 2 → NaF + Cl 2. Using Mole Ratios.


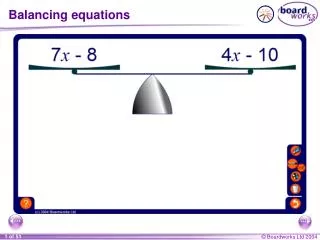
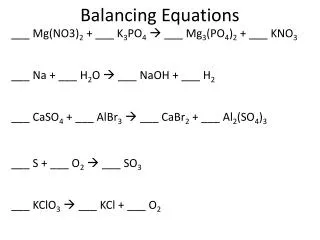
Balancing Equations
E N D
Presentation Transcript
Balancing Equations H2 + Cl2→ HCl
Balancing Equations KI + Br2→ KBr + I2
Balancing Equations CH4 + O2→ CO2 + H2O
Balancing Equations V2O5 + Ca → V + CaO
Balancing Equations NaCl + F2 → NaF + Cl2
Using Mole Ratios In the reaction, 2H2O → 2H2 + O2, if you start with 2 mol of water, how many moles of hydrogen gas are produced?
Using Mole Ratios If you start with 5 mol of O2 in the reaction, 2Mg + O2→ 2MgO, how many moles of Mg will you need?
Using Mole Ratios In the reaction, H2S + 2O2→ H2SO4, the law of definite proportions predicts that for every mole of H2S you will need how many moles of O2?