Understanding Atomic Bonding: Electronegativity, Ionic and Covalent Bonds in Chemistry
This comprehensive review by Korey Tremblay explores the fundamentals of atomic bonding, focusing on key concepts such as protons, electrons, nuclei, and atomic orbitals. It covers the differences between ionic and covalent bonds, emphasizing the concept of electronegativity, the octet rule, and Lewis dot diagrams. The review includes examples, such as sodium chloride (NaCl) and magnesium bromide (MgBr2), to illustrate these principles. Engage in practical experiments like the penny drop and salt experiment, and enhance your understanding with worksheets and illustrations examining bond formation and interactions.

Understanding Atomic Bonding: Electronegativity, Ionic and Covalent Bonds in Chemistry
E N D
Presentation Transcript
Atomic Bonding Korey Tremblay Chemistry
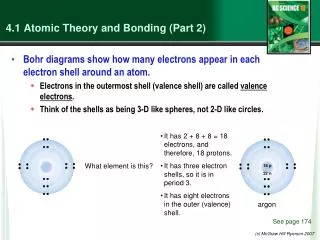
Review Protons and Electrons Nucleus Orbitals Atomic Radius
Goals Ability to answer questions about: Electronegativity Lewis Dot Diagrams Different types of Bonding
Electronegativity Atomic radius Electronegativity number
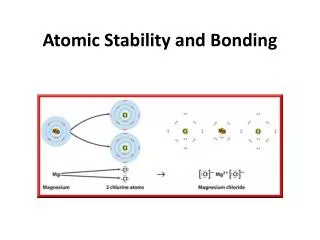
Examples Sodium Chloride – NaCl Magnesium Bromide – MgBr2
Examples Sodium Chloride – NaCl Magnesium Bromide – MgBr2 Notice something about these examples?
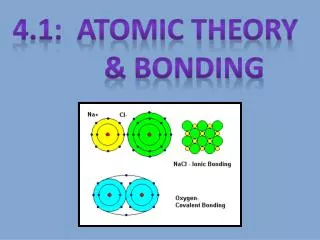
Ionic compounds “Stealing” of electrons Metal and Nonmetal Electronegativity difference Octet rule
Lewis Diagrams – Ionic compounds Electrons belong only to one atom Each atom is charged Lewis diagram of Water Salt experiment
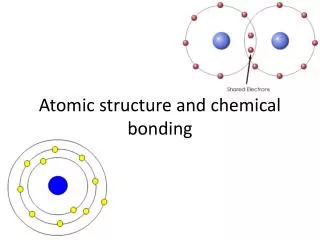
Covalent bonds Both atoms “share” the electrons Forms a stronger bond Octet rule Oxygen
Lewis Diagrams – Covalent bond Drawing a Lewis Diagram for O2 Partial Charges (water) Hydrogen bonds
Hydrogen Bonding Based on attraction of charges No electrons are shared or transferred Penny drop experiment
Next week… VSEPR Like charges repel