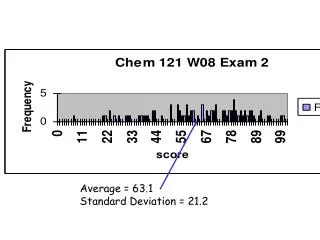
Average = 63.1 Standard Deviation = 21.2
Average = 63.1 Standard Deviation = 21.2. Average = 137.5 = C+ Standard Deviation = 36.3. Approximate Grade based on first 2 exams: A = 186+ A- = 180-185 B+ = 170-179 C = 100-126 B = 155-169 C- = 90-99 B- = 138-154 D/F lower than that C+ = 126-137. Fig. 7.3.

Average = 63.1 Standard Deviation = 21.2
E N D
Presentation Transcript
Average = 63.1 Standard Deviation = 21.2
Average = 137.5 = C+ Standard Deviation = 36.3
Approximate Grade based on first 2 exams: A = 186+ A- = 180-185 B+ = 170-179 C = 100-126 B = 155-169 C- = 90-99 B- = 138-154 D/F lower than that C+ = 126-137
The Spectrum of Electromagnetic Radiation • The wavelength of visible light is between 400 and 700 nanometers • Radio, TV , microwave and infrared radiation have longer wavelengths (shorter frequencies), and lower energies than visible light. • Gamma rays and X-rays have shorter wavelengths (larger frequencies), and higher energies than visible light!
Electromagnetic Radiation c • WAVELENGTH - The distance between identical points on successive waves. ( ) • FREQUENCY - The number of waves that pass through a particular point per second. () • AMPLITUDE - The vertical distance from the midline to a peak, or trough in the wave.
Calculation of Frequency from Wavelength Problem: The wavelength of an x-ray is 1.00 x10 -9 m or 1 nm, what is the frequency of this x-ray? If the wavelength of long-wavelength electromagnetic radiation is 7.65 x 104 m, what is the frequency of this long-wavelength radiation used to contact submerged nuclear submarines at sea? Plan: Use the relationship between wavelength and frequency to obtain the answer. wavelength x frequency = speed of light! Solution: speed of light wavelength(m) frequency(cycles/sec) = 3.00 x 108 m/s 1.00 x 10 - 9 m a) frequency = = 3.00 x 1017 cycles/sec 3.00 x 108 m/s 7.65 x 104 m b) frequency = = 3.92 x 103 cycles/s
The Diffraction Pattern Caused by Light Passing through Two Adjacent Slits Fig. 7.5
The Photoelectric Effect - I • Below the threshold energy, nothing occurs ! • Above the threshold, the kinetic energy of the ejected electrons is proportional to the frequency of the light. • Also, when above the threshold, as intensity of the light increases, so does the number of ejected electrons. • All metals experience this effect, but each has a unique threshold frequency.
The Photoelectric Effect - II • Albert Einstein • Theorized Photons • Won Nobel prize - 1921 • Photons have an energy equal to: E = h • h = Plank’s Constant, and is equal to: 6.6260755 x 10 - 3 4Jsec
Photons and the Photoelectric Effect • Einstein also stated that the change in the photon’s energy was equal to the ejected electron’s energy. • Therefore, the photon’s energy equaled the electron’s kinetic energy added to the electron’s binding energy • ETotal = Electron binding + Electron Kinetic energy
Calculation of Energy from Frequency Problem: What is the energy of a photon of electromagnetic radiation being emitted by radio station KBSG 97.3 FM ( 97.3 x 108 cycles/sec)? What is the energy of a gamma ray emitted by Cs137 if it has a frequency of 1.60 x 1020/s? Plan: Use the relationship between energy and frequency to obtain the energy of the electromagnetic radiation (E = hv). Solution: EKBSG =hv = (6.626 x 10 -34Js)(9.73 x 109/s) = 6.447098 x 10 -24J EKBSG = 6.45 x 10 - 24 J Egamma ray =hv = ( 6.626 x 10-34Js )( 1.60 x 1020/s ) = 1.06 x 10 -13J Egamma ray = 1.06 x 10 - 13J
Calculation of Energy from Wavelength Problem: What is the energy of a photon of electromagnetic radiation that is used in microwave ovens to cook things by rotating water molecules, the wavelength of the radiation is 122 mm. Plan: Convert the wavelength into meters, then the frequency can be calculated using the relationship;wavelength x frequency = C (where C is the speed of light), then using E=hv to calculate the energy. Solution: wavelength = 122 mm = 1.22 x 10 -1m C wavelength 3.00 x108 m/s 1.22 x 10 -1m frequency = = = 2.46 x 1010 cycles/s Energy = E = hv = (6.626 x 10 -34Js)(2.46 x 1010/s) = 1.63 x 10 - 23 J
Photoelectric Effect -I • Mininum energy to remove an electron from potassium metal is 3.7 x 10 -19J. Will photons of frequencies of 4.3 x 1014/s (red light) and 7.5 x 1014 /s (blue light) trigger the photoelectric effect? • E red = hv = (6.626 x10 - 34Js)(4.3 x1014 /s) E red = 2.8 x 10 - 19 J • E blue = hv = (6.626 x10 - 34Js)(7.5x1014 /s) E blue = 5.0 x 10 - 19 J
Photoelectric Effect - II • Since the binding energy of potassium is = 3.7 x 10 - 19 J • The red light will not have enough energy to knock an electron out of the potassium, but the blue light will eject an electron ! • E Total = E Binding Energy + EKinetic Energy of Electron • E Electron = ETotal - E Binding Energy • E Electron = 5.0 x 10 - 19J - 3.7 x 10 - 19 J = 1.3 x 10 - 19Joules
Light and Atoms • When an atom gains a photon, it enters an excited state. • This state has too much energy - the atom must lose it and return back down to its ground state, the most stable state for the atom. • An energy level diagram is used to represent these changes.
The Line Spectra of Several Elements Hg Fig. 7.8
Energy Level Diagram • Energy Excited States photon’s path Ground State Light Emission Light Emission Light Emission
Figure 7.10: Energy-level diagram for the electron in the hydrogen atom.
Figure 7.11: Transitions of the electron in the hydrogen atom.
Emission Energetics - I Problem: A sodium vapor light street light emits bright yellow light of wavelength = 589 nm. What is the energy change for a sodium atom involved in this emission? How much energy is emitted per mole of sodium atoms? Plan: Calculate the energy of the photon from the wavelength, then calculate the energy per mole of photons. Solution: ( 6.626 x 10 -34J s)( 3.00 x 10 8m/s) h x c wavelength Ephoton = hv = = 589 x 10 -9m Ephoton = 3.37 x 10 -19J Energy per mole requires that we multiply by Avogadro’s number. Emole = 3.37 x 10 -19J/atom x 6.022 x 1023 atoms/mole = 2.03 x 105 J/mol Emole = 203 kJ / mol
Emission Energetics - II Problem: A compact disc player uses light with a frequency of 3.85 x 1014 per second. What is this light’s wavelength? What portion of the electromagnetic spectrum does this wavelength fall? What is the energy of one mole of photons of this frequency? Plan: Calculate the energy of a photon of the light using E=hv, and wavelength x C = v . Then compare the frequency with the electromagnetic spectrum to see what kind of light we have. To get the energy per mole, multiply by Avogadro’s number. Solution: 3.00 x 108m/s 3.85 x 1014/s wavelength = c / v = = 7.78 x 10 -7 m = 778 nm 778 nm is in the Infrared region of the electromagnetic spectrum Ephoton = hv = (6.626 x 10 -34Js) x ( 3.85 x 1014 /s) = 2.55 x 10 -19 J Emole = (2.55 x 10 -19J) x (6.022 x 1023 / mole) = 1.54 x 105 J/mole
1 n12 1 n22 1 2 2 1 4 2 Using the Rydberg Equation Problem: Find the energy change when an electron changes from the n=4 level to the n=2 level in the hydrogen atom? What is the wavelength of this photon? Plan: Use the Rydberg equation to calculate the energy change, then calculate the wavelength using the relationship of the speed of light. Solution: Ephoton = -2.18 x10 -18J - = Ephoton = -2.18 x 10 -18J - = - 4.09 x 10 -19J h x c E (6.626 x 10 -34Js)( 3.00 x 108 m/s) wavelength = = = 4.09 x 10 -19J wavelength = 4.87 x 10 -7 m = 487 nm
Wave Motion in Restricted Systems Fig. 7.12
The de Broglie Wavelengths of Several Objects Substance Mass (g) Speed (m/s) (m) Slow electron 9 x 10 - 28 1.0 7 x 10 - 4 Fast electron 9 x 10 - 28 5.9 x 106 1 x 10 -10 Alpha particle 6.6 x 10 - 24 1.5 x 107 7 x 10 -15 One-gram mass 1.0 0.01 7 x 10 - 29 Baseball 142 25.0 2 x 10 - 34 Earth 6.0 x 1027 3.0 x 104 4 x 10 - 63 Table 7.1 (p. 274)
Figure 7.18: Plot of y2 for the lowest energy level of the hydrogen atom.
Figure 7.19: Probability of finding an electron in a spherical shell about the nucleus.
Figure 7.20: Scanning tunneling microscope of benzene molecules on a metal surface.Photo courtesy of IBM Almaden Research Center.
Figure 7.22: Quantum corral.Photo courtesy of IBM Almaden Research Center; research done by Dr. Don Eigler and co-workers.
Figure 7.24: Cross-sectional representations of the probability distributions of S orbitals.
Figure 7.25: Cutaway diagrams showing the spherical shape of S orbitals.
Light Has Momentum • momentum = p = mu = mass x velocity • p = Plank’s constant / wavelength • or: p = mu = h/wavelength • wavelength = h / mu de Broglie’s equation • de Broglie’s expression gives the wavelength relationship of a particle traveling a velocity = u !!
de Broglie Wavelength Calc. - I Problem: Calculate the wavelength of an electron traveling 1% of the speed of light ( 3.00 x 108m/s). Plan: Use the de Broglie relationship with the mass of the electron, and its speed. Express the wavelength in meters and nanometers. Solution: electron mass = 9.11 x 10 -31 kg velocity = 0.01 x 3.00 x 108 m/s = 3.00 x 106 m/s h m x u 6.626 x 10 - 34Js ( 9.11 x 10 - 31kg )( 3.00 x 106 m/s ) wavelength = = = kg m2 s2 J = therefore : wavelength = 0.24244420 x 10 - 9 m = 2.42 x 10 -10 m = 0.242 nm
Heisenberg Uncertainty Principle • It is impossible to know simultaneously both the position and momentum (mass X velocity) and the position of a particle with certainty !