Understanding Energy and its Forms: A Comprehensive Overview
Explore the concept of energy, Newton’s Laws, forms of energy, heat transfer, thermodynamics laws, and applications. Learn about kinetic and potential energy, specific heat, temperature scales, and more. Dive into how energy works in various systems and the efficiency of energy conversions.

Understanding Energy and its Forms: A Comprehensive Overview
E N D
Presentation Transcript
Newton’s Laws • An object in motion will stay in motion (or rest at rest) unless acted on by an outside force. (Inertia) • Force = mass x acceleration (Units: Newtons = kg x m/s2) • For every action there is an equal and opposite reaction.
Work • Energy = ability to do work • Work = force exerted over a distance • W (or “energy”) = F xd • 1 Joule = 1 Newton x 1 meter • 4.18 Joule = 1.00 calorie
Power • Rate at which work is done • Power = Work / time • Watts = Joules / seconds • SO… Energy can also be expressed this way: • Energy = Power x time • Joules = Watts x seconds
Consider: • What does it mean to give electrical appliances a rating in watts? • What is a kilowatt? What is a killowatt-hour? • Why do we describe food in terms of calories?
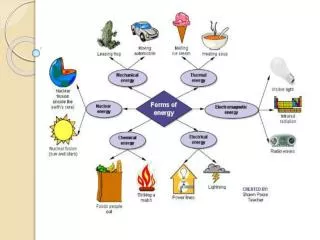
Forms of Energy • Kinetic energy = Energy of moving objects • KE = ½ mass x velocity2 • KE = ½ mv2 • Joules = ½ kg x (m/s)2 • Potential energy = Energy stored due to position or composition • PE = mass x acceleration due to gravity x height • PE = mgh • Joules = kg x m/s2xm
Consider: • How do each of the following represent KE? • Heat radiating from a car • The sound of my voice • Muscles contracting • Light traveling to your eye
Consider: • How do each of the following represent potential energy? • A book on the edge of a table • A slice of pizza • Two magnets near each other • An electrical outlet • A stretched rubber band
What is heat? • Kinetic energy caused by atoms moving and colliding • Flows from warm body to cold body • Not the same as temperature
Temperature Scales • ° F = (1.8 x ° C) + 32 • ° C = (° F – 32) / 1.8 • Both use freezing and boiling points of water as standards • Kelvin = ° C – 273 • 0 K = absolute zero
Specific Heat • Ability of a material to absorb/lose heat • Q = mcΔt • Heat gained/lost = mass x specific heat x change in temp • If two substances have equal mass and equal change in temp., what does the amount of heat gained depend on?
Heat transfer • Conduction – transfer due to atoms colliding or vibrating • Convection – transfer due to bulk movement of a fluid (gas or liquid) • Radiation – transfer due to electromagnetic radiations (uses electrical/magnetic fields instead of matter) • file:///Users/johnb/Desktop/lsps07_int_heattransfer/lsps07_int_heattransfer.html • How can these types of transfer work together?
1st Law of Thermodynamics:Conservation of Energy • Energy cannot be created or destroyed but can be transformed • PE = KE • mgh = ½ mv2 • How can our examples of PE be converted into KE? Is the reverse also true? • Are energy conversions 100% efficient?
2nd Law of Thermodynamics: Entropy • Statements: • Heat will not flow spontaneously from a cold to a hot body. • You cannot construct an engine that does nothing but convert heat to useful work. • Every isolated system becomes more disordered with time. (Entropy) http://www.stolaf.edu/people/giannini/flashanimat/transport/diffusion.swf
Applications • Can any system transfer energy with 100% efficiency? What does this mean in living systems? • How do we maintain order in systems (especially living systems)? • Where does energy go? Is it truly “lost”?
Back to the Sun – Nuclear Fusion • High pressure and high temperature force atoms together • Mass converted to energy • Energy radiates into space • How do atoms relate to energy?