Ch 8: Introducing Acids and Bases
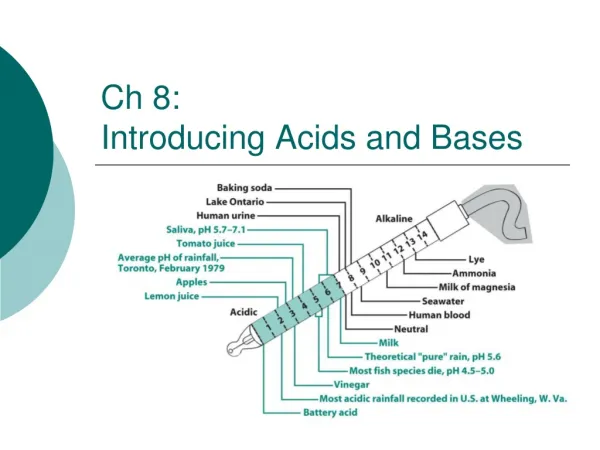
Ch 8: Introducing Acids and Bases. pH of precipitation in the United States 2001, and in Europe as reported in 2002. What are Acids and Bases?. acid = substance that increases the concentration of H 3 O + base = decreases the concentration of H 3 O + (by increasing the amount of OH - ).

Ch 8: Introducing Acids and Bases
E N D
Presentation Transcript
pH of precipitation in the United States 2001, and in Europe as reported in 2002.
What are Acids and Bases? acid = substance that increases the concentration of H3O+ base = decreases the concentration of H3O+ (by increasing the amount of OH-)
Bronsted-Lowry Acid-Base Theory acid = proton donor (H+) base = proton acceptor HCl + H2O → HCl + NH3→
Relation Between [H+], [OH-], and pH H2O + H2O H3O+ + OH- H2O H+ + OH- Kw = [H+][OH-] = 1.01 x 10-14 at 25 oC equivalent
Example, p. 169: Concentration of H+ and OH- in Pure Water at 25 oC Calculate the concentrations of H+ and OH- in pure water at 25 oC.
As the concentration of H+ increases, OH- must decrease and vica-versa Example, p. 169: Finding [OH-] when H+ is Known. What is the concentration of OH- if [H+] = 1.0 x 10-3 M at 25 oC?
pH - a measure of the aciity of a solution ("puissance d'hydrogen") pH = -log [H+] (approximately!) [H+] = 10-3 M [H+] = 10.0 M [H+] = 10-10 M
Strengths of Acids and Bases strong =complete (100%) dissociation MEMORIZE these strong acids and bases - all other acids and bases are weak
HA H+ + A- OR HA + H2O H3O+ + A- B + H2O BH+ + OH- weak = incomplete dissociation
carboxylic acids = weak acids amines = weak bases RNH2 primary R2NH secondary R3N tertiary : : : Classes of Weak Acids and Bases polyprotic acids and bases H2CO3 CO32- H3PO4 PO43- Ca(OH)2
A- + H2O HA + OH- Relation Between Ka and Kb HA + H2O H3O+ + A- salt = conjugate baseundergoes hydrolysis
Example, p. 174: Ka for acetic acid is1.75 x 10-5. Find Kb for the acetate ion.
pH of solutions of strong acids and bases HA → H+ + A- BOH → B+ + OH- H2O H+ + OH- [H+] = [OH-] = 1.0 x 10-7 strong acids and bases completely dissociate Case I: concentration of acid or base >> 10-7
pH of a strong acid: Example p. 175 Find the pH of 4.2 x 10-3 M HClO4
Case II: concentration of acid or base 10-7 Now the contribution of H+ from water must be included -
pH of a strong base at a low concentration: “trick question” top of p. 176 Find the pH of 4.2 x 10-9 M KOH
pH of solutions of weak acids and bases (Sec 8-6, 8-7) – the “ICE” table Calculate the pH of a 0.020 M benzoic acid solution. Ka = 6.28 x 10-5 I: Exact solution using quadratic equation
Weak Base Equilibrium, Example p. 183 Find the pH of a 0.0372 M solution of the commonly encountered (?) weak base cocaine. Kb = 2.6 x 10-6
Ch 9:Buffers buffer = resists changes in pH; solution of a weak acid or base and their salts
Henderson-Hasselbach Equation derivation and assumptions:
Example, p.191: Using the H-H Equation Sodium hypochlorite (NaOCl) was dissolved in a solution buffered to pH = 6.20. Find the ratio [OCl-]/[HOCl]
Example, p. 192: A Buffer Solution Find the pH of a solution prepared by dissolving 12.43 g of TRIS (FM = 121.136) plus 4.67 g TRIS hydrochloride (FM = 157.597) in 1.00 L of water.
How to Prepare a Buffer Solution • Consult a table of pKa's and pick the weak acid or base closest to the pH you need. • Solve for the ratio mol salt/(mol acid/base) • Choose a reasonable value for either mol salt or mol acid/base and solve for the other • After preparing the buffer, adjust the pH to the desired value (you never get exactly what you calculate because of the assumptions made in deriving the H-H equation
Buffer Capacity: How well a solution resists changes in pH when an acid or base is added: when the pH = pKa!
Example, p.198 HA = H+ + A- mol A- = 0.0383, mol HA = 0.9617