Accuracy and Precision
650 likes | 2.26k Vues
Accuracy and Precision. To many people, accuracy and precision mean the same thing: to someone involved in measurement, the two terms should have very different meanings.

Accuracy and Precision
E N D
Presentation Transcript
Accuracy and Precision To many people, accuracy and precision mean the same thing: to someone involved in measurement, the two terms should have very different meanings. One way in which this distinction is apparent is the difference between a poll (a measurement) and a vote (a count). The result obtained from counting will be an exact result (barring blunder), while the result obtained from measuring will only approach the truth.
We are taught how to deal with exact numbers, but are sometimes not aware of the nature of results we obtain from approximated or measured values.
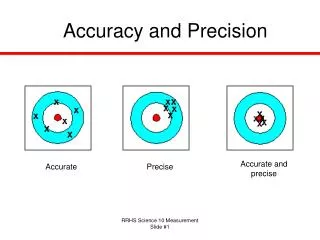
The precision of an instrument reflects the number of significant digits in a reading; Precision indicates how close together or how repeatable the results are. A precise measuring instrument will give very nearly the same result each time it is used.
The accuracy of an instrument reflects how close the reading is to the 'true' value measured. Accuracy indicates how close a measurement is to the accepted value. For example, we'd expect a balance to read 100 grams if we placed a standard 100 g weight on the balance. If it does not, then the balance is inaccurate
There is no such thing as a Perfect Measurement As a consequence of the above fact, all measurements should include an estimate of the accuracy conveyed by a given reading. The accuracy estimate is reflected in the error term. Result = Every measurement should include the value, an error term and the units.
Precise and Accurate Accurate, Not Precise Neither Precise Nor Accurate Precise, Not Accurate
Significant Figures Significant figures are the number of reliably known digits used to locate a decimal point reported in a measurement. Proper use of significant figures ensures that you correctly represent the uncertainty of your measurements. For example, scientists immediately realize that a reported measurement of 1.2345 m is much more accurate than a reported length of 1.2 m.
Guidelines for Significant Figures: When a measurement is properly stated in scientific notation all of the digits will be significant. For example: 0.0035 has 2 significant figures which can be easily seen when written in scientific notation as 3.5 x 10-3. Fortunately, there are a few general guidelines that are used to determine significant figures: 1. Whole Numbers 2. Integers and Defined Quantities 3. Multiplication and Division 4. Addition and Subtraction
Whole Numbers: The following numbers are all represented by three significant digits. Note that zeros are often place holders and are not significant. 0.00123 0.123 1.23 12.3 123 12300 (The zeros here often cause confusion. As written here, the zeros are not significant. If they were, in fact, significant, then the use of scientific notation would remove all ambiguity and the number would be written 1.2300 x 104.)
The following numbers are all represented by one significant digit. 0.005 0.5 5 500 5,000,000
The following numbers are all represented by four significant figures. 0.004001 0.004000 40.01 40.00 4321 432.1 43,210,000
PERCENT ERROR The significant figures rule in only approximate, and in some cases may underestimate the precision of the answer. Turn to page 7 in your physics book Use the significant figures rule, but consider the % uncertainty too, and add an extra digit if it gives a more realistic estimate of uncertainty
Integers and Defined Quantities: Integers are assumed to have an infinite number of significant figures. For example, the 2 in C = 2pr, is exactly two and we can assume that the number has an infinite number of significant figures. However, the conversion factor 2.54 cm which is used to convert inches to centimeters has three significant figures.
Multiplication and Division: When multiplying or dividing numbers, the result should have only as many significant figures as the quantity with the smallest number of significant figures being used in the calculation. For example, with your calculator multiply 4.7 and 5.93. The calculator returns 27.871 as the answer. A common mistake students make is to record what comes out of the calculator as the correct answer. However, since 4.7 has only 2 significant figures, the result must be truncated to 2 significant figures as well. Taking all this into account and remembering to round appropriately, the result should be reported as 28.
Addition and Subtraction: RULE: When adding or subtracting your answer can only show as many decimal places as the measurement having the fewest number of decimal places. 3.76 g + 14.83 g + 2.1 g = 20.69 g = 20.7g
SCIENTIFIC NOTATION Precision as to the significance of a value may be avoided by expressing the number in standard exponential notation.
Addition and Subtraction When adding or subtracting numbers in scientific notation, their powers of 10 must be equal. If the powers are not equal, then you must first convert the numbers so that they all have the same power of 10. (6.7 x 109) + (4.2 x 109) = (6.7 + 4.2) x 109 = 10.9 x 109 = 1.09 x 1010 (6.7 x 109) + (4.2 x 107) = (670 x 107) + (4.2 x 107) So to make the exponent lower move the decimal of the number to the right or to make the exponent larger move the decimal of the number to the left. Then you can add or subtract.
Multiplication and Division It is very easy to multiply or divide just by rearranging so that the powers of 10 are multiplied together (6 x 102) x (4 x 10-5) = (6 x 4) x (102 x 10-5) = 24 x 102-5 = 24 x 10-3 = 2.4 x 10-2. (8 x 102) = 8/4 x 102-5 = 2 x 10-3 (4 x 105)
ADDITIONAL PRINCIPLES PRINCIPLE NUMBER ONEIf you are using exact constants, such as thirty-two ounces per quart or one thousand milliliters per liter, they do not affect the number of significant figures in you answer. For example, you might need to calculate how many feet equal 26.1 yards. The conversion factor you would need to use, 3 ft/yard, is an exact constant and does not affect the number of significant figures in your answer. Therefore, 26.1 yards multiplied by 3 feet per yard equals 78.3 feet which has 3 significant figures.
PRINCIPLE NUMBER TWOIf you are using constants which are not exact (such as pi = 3.14 or 3.142 or 3.14159) select the constant that has at least one or more significant figures than the smallest number of significant figures in your original data. This way the number of significant figures in the constant will not affect the number of significant figures in your answer. For example, if you multiply 4.136 ft which has four significant figures times pi, you should use 3.1416 which has 5 significant figures for pi and your answer will have 4 significant figures.
PRINCIPLE NUMBER THREEWhen you are doing several calculations, carry out all of the calculations to at least one more significant figure than you need. When you get the final result, round off. For example, you would like to know how many meters per second equals 55 miles per hour. The conversion factors you would use are: 1 mile equals 1.61 x 103 meter and 1 hour equals 3600 seconds. Your answer should have two significant figures. Your result would be 88.55 divided by 3600 which equals 24.59 m/sec. This rounds off to 25 m/sec. By carrying this calculation out to at least one extra significant figure, we were able to round off and give the correct answer of 25 m/sec rather than 24 m/sec.
How many significant figures are in each of the following numbers?a) 1.234b) 1.2340c) 1.234 x 10-3d) 1.2340 x 10-3e) 1234f) 12340g) 0.012340 a) 1.234: 4b) 1.2340: 5c) 1.234 x 10-3: 4d) 1.2340 x 10-3: 5e) 1234: 4f) 12340: 4 or 5g) 0.012340: 5
Complete the following operations and express the answer with the correct number of significant figures:a) 1.421+ 0.4372 =b) 0.0241 + 0.11 =c) 0.14 + 1.2243 =d) 760.0 + 0.011 =e) 1.0123 - 0.002 =f) 123.69 - 20.1 =g) 463.231 - 14.0 =h) 47.2 - 0.01 = a) 1.421+ 0.4372 = 1.858b) 0.0241 + 0.11 = 0.13c) 0.14 + 1.2243 = 1.36d) 760.0 + 0.011 = 760.0e) 1.0123 - 0.002 = 1.010f) 123.69 - 20.1 = 103.6g) 463.231 - 14.0 = 449.2h) 47.2 - 0.01 = 47.2
Perform the indicated operations. Express your answers with the correct number of significant figures:a) 42.3 x 2.61 =b) 0.61 x 42.1 =c) 46.1 / 1.21 =d) 23.2 / 4.1 = a) 42.3 x 2.61 = 110b) 0.61 x 42.1 = 26c) 46.1 / 1.21 = 38.1d) 23.2 / 4.1 = 5.7
http://www.carlton.paschools.pa.sk.ca/chemical/Sigfigs/accuracy_and_precision.htmhttp://www.carlton.paschools.pa.sk.ca/chemical/Sigfigs/accuracy_and_precision.htm http://honolulu.hawaii.edu/distance/sci122/SciLab/L5/accprec.html