Overview of Atomic Energy for Boy Scouts: Merit Badge Workshop
Join us for an engaging workshop designed to help Boy Scouts earn their Atomic Energy Merit Badge. This session will cover key concepts like atomic structure, isotope definitions, fission vs. fusion, and the effects of radiation. Scouts will participate in interactive stations exploring the history of the atom and its applications in nuclear energy. The workshop includes a lunch break and an opportunity to complete hands-on activities. Prepare to learn about radiation safety and its implications in today’s world.


Overview of Atomic Energy for Boy Scouts: Merit Badge Workshop
E N D
Presentation Transcript
Atomic Energy Overview Boy Scout Merit Badge 2006 American Nuclear Society
Schedule • 9:00-9:55 Introduction • 10:00-10:40 Station 1 • 10:45-11:25 Station 2 • 11:30-12:10 Lunch • 12:15-12:55 Station 3 • 1:00-1:40 Station 4 • 1:45-2:25 Station 5 • 2:30-3:00 Wrap-Up/Sign Blue Cards
Blue Cards Put my name in appropriate spot Workshop Leader Address Line 1 Address Line 2 Phone Number
Requirements 1a. Radiation Effects 1b.Drawing of radiation symbol 2. Definitions 3. Scientists 4a. Model of atoms 4b. Fission pictures 4c. Critical Mass 5c. Time, distance and shielding 5f. X-ray facility 5g. Cloud Chambers 6b. US nuclear reactors 7. Nuclear Careers
Brief History of the Atom • 500 BC Democritus Atom • Long time (Romans Dark Ages) • 1808 AD Dalton Plum Pudding • 1911 Rutherford Nucleus • 1913 Bohr Orbits • 1920’s Many People Quantum Mechanics
So What is an Atom? • Atoms are made up of protons, neutrons & electrons • Protons: + charge • Neutrons: no charge • Electrons: - charge • Atoms want to have no net charge • #protons = #electrons
Mass of an Atom • Masses • Proton: 1 amu • Neutron: 1 amu • Electron: .000549 amu • So mass of atom ~ • #neutrons + #protons
What is the mass of: • Helium? • 2 protons, 2 neutrons, 2 electrons • Oxygen • 8 protons, 8 neutrons, 8 electrons • Fissile Uranium • 92 protons, 143 neutrons, 92 electrons • Alpha particle • 2 protons, 2 neutrons
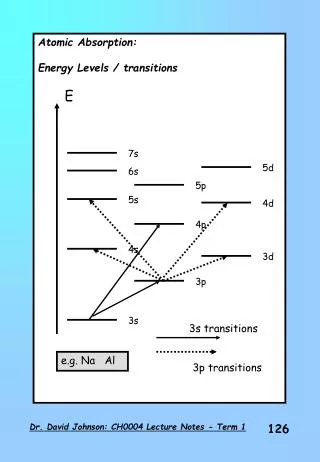
Isotopes • Isotopes are similar elements with different amounts of neutrons • There are similar properties • Some have very different properties • Stable and unstable versions of atoms
Example • Sodium – 23 • 11 protons, 12 neutrons • Very plentiful, in salt that you eat (NaCl) • Sodium – 24 • 11 protons, 13 neutrons • Not natural • Highly radioactive, beta decay • Used to find leaks in industrial pipes
Put together models of: • Hydrogen • 1 Proton, 1 electron • Deuterium • 1 Proton, 1 neutron, 1 electron • Tritium • 1 Proton, 2 neutrons, 1 electron • What are the atomic and mass numbers of each isotope? • What are the net charges? • (Requirement 4a)
Fission vs. Fusion • Fission • Makes 20% of our electricity • Breaking apart of Heavy Nuclei • Nuclear Reactor tour later Today! • Example: • N + U-235 Ba-139 + Kr-94 + 3N • Fusion • Powers the Sun • Hard to get on Earth • Combination of Light Nuclei • IEC Fusion Tour later Today! • Example: • H-3 + H-2 N + He-4 • Tritium + Deuterium neutron + Helium
Where does radiation come from? • The sun • Soil, water and vegetation • Internal sources • Potassium-40 (bananas) • Carbon-14 (air) • Lead-210 (radon) • Man-made sources • Medical sources (x-rays, radiation…) • Nuclear Power
Types of Radiation Ionizing radiation • Produces ions in the material it strikes • Non-ionizing radiation • Can cause damage by physically striking material
Ionizing radiation • Alpha particles • Beta particles • Photons • Gamma rays • X-rays
Non-Ionizing Radiation • Low energy photons • Such as light, infrared light, etc • Neutrons • Neutral particle in nucleus • Like a proton but with no charge
Radioactive Material • What is it? • Material that releases radiation. • How do we get rid of it? • When an atom decays, it may or may not be radioactive still. Eventually, it will decay to a stable atom
Decay • Radioactive material has a probability of decaying • Overtime, this probability averages into a half life • Half-life: Amount of time it takes for half of the radioactive substance to decay
Biological Effects of Radiation • Deterministic • responses which increase in severity with increased dose (sunburn) • Stochastic • effects which have an increased probability of occurrence with increased dose, but whose severity is unchanged (skin cancer)
Radiation Exposure • To avoid accidental exposure, we use the radiation hazard symbol • Colors • Magenta or Black • Yellow Background • Color one of your own • Why must people use radioactive materials carefully? (Requirement 1a and 1b)