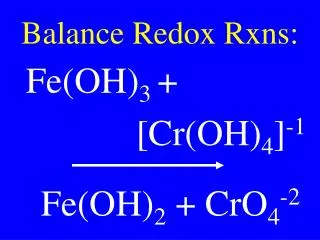Topic: Redox
Topic: Redox. Aim: How do we find the oxidation numbers of an element? Do Now: What are the possible oxidation #s of gold? Neon? Mg? Cl? HW: finish ox states wkst. Assigning Oxidation States. Oxidation states/ numbers - used to identify what has been oxidized and reduced in a rxn

Topic: Redox
E N D
Presentation Transcript
Topic: Redox Aim: How do we find the oxidation numbers of an element? Do Now: What are the possible oxidation #s of gold? Neon? Mg? Cl? HW:finish ox states wkst
Assigning Oxidation States • Oxidation states/numbers - used to identify what has been oxidized and reduced in a rxn • Oxidation state/number • # assigned to keep track of e- gained or lost during redox • particular charge of an element • Found in top right corner of box on Periodic Table
Rules for Assigning Oxidation #s • Free elements (not combined with others) have an oxidation number of 0. • Ex: Na, Cl2 • In a compound, the sum of the oxidation numbers is0. • The overall charge of the entire compound is 0. • Ex: NaCl KClO3 MgCl2 HClO4 N2O
Rules, cont. • All Group 1 metals have an oxidation number of +1 (when in compounds). • All Group 2 metals have an oxidation number of +2 (when in compounds). • Fluorine always has an oxidation number of -1 in all compounds.
Rules, cont. • Hydrogen has an oxidation number of +1 in all compounds • Except: In metal hydrides (metal – like from Groups 1 & 2 – plus hydrogen) Ex: LiH, CaH2 • Oxygen has an oxidation number of -2 in all compounds • Except: In peroxides Ex: H2O2, Na2O2, K2O2 • In compounds with fluorine Ex: OF2
Rules, cont. • In simple ions (with only one type of atom), the oxidation number equals the charge of the ion. • Ex: Na+ (+1), Cl- (-1), • In polyatomic ions (Table E), the sum of the oxidation numbers of all the atoms must equal the charge of the ion. • Ex: SO42- (-2), PO43- (-3), HSO4- (-1)
sodium nitrate ____________________________________ • ammonia ____________________________________ • zinc oxide ____________________________________ • water ____________________________________
calcium hydride ____________________________________ • carbon dioxide ____________________________________ • nitrogen ____________________________________
sodium sulfate ____________________________________ • aluminum hydroxide ____________________________________ • magnesium phosphate ____________________________________