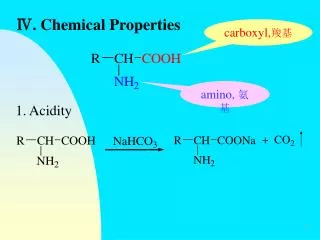
Chemical Properties
Realplayer videosJCE Software Chemistry Comes Alive!.mov. Chemical Properties. Section 17.3. Demo - Candle. What physical changes can be made to each item? Candle Match Stick Light the match and put it near the side of the candle. Is the melting wax a physical or a chemical change?

Chemical Properties
E N D
Presentation Transcript
Realplayer videos\JCE Software Chemistry Comes Alive!.mov Chemical Properties Section 17.3
Demo - Candle • What physical changes can be made to each item? • Candle • Match • Stick • Light the match and put it near the side of the candle. Is the melting wax a physical or a chemical change? • Light the candle. As the wax and the wick burn, is it a physical or a chemical change?
Chemical Properties of Matter • Chemical properties describe a substance’s ability to change into new matter with new properties. Realplayer videos\Video pedia World - Elements Of Chemistry Chemical Reactions2.flv • Flammability: describes whether substances will react in the presence of oxygen and burn when exposed to a flame
carondelet.net Chemical Properties • Reactivity: describes the ability of two or more substances to combine to form one or more new substances. • Rusting – reactivity with oxygen • Explosive reaction of elemental sodium with chlorine to form table salt Realplayer videos\Bill Nye The Science Guy on Chemical Reactions (Full Clip) - Bing Videos.flv Realplayer videos\Reaction of Sodium Chlorine (with subtitles).flv www.artmetal.com
Chemical Property Applications • Galvanized steel is steel coated with zinc to prevent rusting (buckets, nails). • Steel plated with tin was once used in food cans and containers. Today, aluminum cans have replaced most steel cans.
Interesting Facts • Before the early 1960s, there was a group of gases (named noble gases) that were thought to be unreactive. However, in 1962, Dr. Neil Bartlett of Univ. of Brit. Columbia formed a new compound by combining platinum, fluorine, and the noble gas xenon. • In 2002, chemists created new compounds by bonding uranium with argon, krypton and xenon (all noble gases). www2.chemistry.msu.edu
Interesting Facts • Chromium has been used as a coating on car bumpers because it is nonreactive with oxygen (so the bumper is protected from rusting). • Rustproofing involves treating the car’s underside and panels with sealants, which penetrate the seams, cracks, and holes to keep out air and moisture – which would increase the rate of rusting.
Substance Identification • Characteristic properties are used to identify and classify substances. They can include both physical and chemical properties. Examples: density, solubility, flammability, reactivity • These properties do not depend upon the size of the sample (so mass, weight and volume would not be characteristic properties).
Substance Identification • Example: How can you tell the difference between baking soda and baking powder if they are out of their container? • Reactivity with vinegar: • Baking soda reacts • Baking powder does not Realplayer videos\SloMo - Baking soda + vinegar reaction @ 420 fps - Bing Videos.flv
Chemical Changes • Chemical changes are the processes by which one or more substances are changed into new substances.
Indicators of Chemical Change • Change in color or scent Realplayer videos\Chemical Changes color change.mov • Production of a gas – fizzing, foaming, effervescence. Realplayer videos\Chemical Changes gas production.mov
Indicators of Chemical Change • Production of a solid (precipitate) Realplayer videos\Precipitate formation Chemistry Comes Alive!.mov • Production of heat • Production of light or sound Realplayer videos\Heat released in chemical reactions Chemistry Comes Alive!.mov • What indications of chemical change do you see in the next video? Realplayer videos\Indicators of chemical change.flv
Chemical Changes • Matter’s identity is changed in a chemical reaction. • Reversing a chemical reaction (getting back what you started with) is not always possible. If it is possible, it requires more chemical changes to take place. • Could you reverse the explosion of fireworks?
Physical Changes • Since physical changes don’t change the composition of a substance, they are frequently easily reversible. • Example: Heating an ice cube to make it melt e cool the resulting liquid to make it freeze • Dissolving a solid in a liquid emaking the substance recrystallize from the solution Realplayer videos\Crystallization of supersaturated sodium thiosulfate - Bing Videos2.flv
References: • http://www2.chemistry.msu.edu/portraits/PortraitsHH_Detail.asp?HH_LName=BartlettN • http://www.kenyacarbazaar.com/blog/?p=899 • http://carondelet.net/Aspring08/Project2/Caitlin/New%20York%20City.htm • http://www.artmetal.com/images/sculpture_gallery/josh/2007/07/rusted_sculptures_update_0 • http://www.manufactum.com/Artikel/99272/2GalvanizedSteelPlantTub.html • http://besbryant.sharpschool.com/cms/One.aspx?portalId=839802&pageId=2630106 • http://mbt6644.k12.sd.us/frontpage1/project1/inthelab1/pictures.htm • http://miamirealestatecafe.com/2009/12/28/cleaning-tips-for-your-miami-home-using-everyday-items • http://www.bbc.co.uk/food/baking_powder • http://www.etftrends.com/2010/07/price-impact-aluminum-etf/ • http://www.icollector.com/Lot-Of-7-Tin-Cans-Texaco-Capella-Oil-Tin-Can-Transmi_i8458786 • http://qwickstep.com/search/chemical-changes-examples.html • http://www.chemistryland.com/CHM130W/04-Properties/PropertiesBook/ChangesAssignment.htm • http://www.queenmaryschool.net/ScienceFair/?tag=/chemical-change • http://picsdigger.com/keyword/chemical%20changes/ • http://jchemed.chem.wisc.edu/JCESoft/CCA/pirelli/pages/cca1NaIHgCl2.html • http://www.treklens.com/gallery/North_America/Canada/photo154072.htm • http://micro.magnet.fsu.edu/moviegallery/chemicalcrystals.html