Chemical Properties
Chemical Properties. BY: BRIANNA SHIELDS. DO NOW. On the last page of your note packet from last class: Label the arrows on the diagram with the words evaporation, melting, freezing and condensation Also, label where the solids, liquids and gases exist Work with your group members. GOAL.


Chemical Properties
E N D
Presentation Transcript
Chemical Properties BY: BRIANNA SHIELDS
DO NOW • On the last page of your note packet from last class: Label the arrows on the diagram with the words evaporation, melting, freezing and condensation • Also, label where the solids, liquids and gases exist • Work with your group members
GOAL • To distinguish between physical and chemical changes
Chemical Changes Changing the substance into a new one by changing atoms
Other Chemical Indicators 1. Burning 2. Foaming/Fizzing/ Bubbling
Other Chemical Indicators 3. Odor change 4. Color change 5. Corroding Acids
Other Chemical Indicators 6. Rusting metal (iron + oxygen = rust) 7. Explosions (fireworks) 8. Verdigris- greened statutes (copper + sulfur)
Other Chemical Indicators 9. Cooking & some food production (cake baking, cheese production) 10. Chemical processes in nature (photosynthesis)
Chemical Change A substance changes into a new and different substance The atoms are altered Often involves chemical rx Physical and Chemical Changes
Foaming (gas formation) Ex: antacid in water
Corrosion due to acids (bubbling or fizzing)
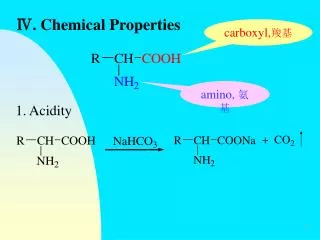
ACIDS Corrosive Sour taste pH paper 1-6 (reddish) Turns litmus red Phenopthalein in acid (no color change) Vinegar & lemon juice BASES Corrosive, slippery Bitter taste pH paper 8-14 (bluish) Turns litmus blue Phenopthalein in base turns pink Soaps, cleaning products 2 chemical indicators
Acids & Bases • Watch the demonstrations at the front counter- are the substances acids or bases? How do you know?
Physical or Chemical Change? • Painting a statue • Chopping wood • Crushing sugar crystals • Dissolving salt • Boiling water • Exploding firecrackers • Mixing baking soda & vinegar
Physical or Chemical Change? • Rotting eggs • Crumpling wax paper • Slicing cucumber • Fizzing antacid in water • Lighting a match • Zipping a coat up • Rusting bike chains