Chapter 10 Catalytic Reforming and Isomerization
340 likes | 2.94k Vues
Chapter 10 Catalytic Reforming and Isomerization. The demand of today’s automobiles for high-octane gasolines has stimulated the use of catalytic reforming

Chapter 10 Catalytic Reforming and Isomerization
E N D
Presentation Transcript
Chapter 10Catalytic Reforming and Isomerization • The demand of today’s automobiles for high-octane gasolines has stimulated the use of catalytic reforming • In catalytic reforming, the change in the boiling point of the stock passed through the unit is relatively small as the hydrocarbon molecular structures are rearranged to form higher-octane aromatics with only a minor amount of cracking. • Thus catalytic reforming primarily increases the octane of motor gasoline rather than increasing its yield; in fact, there is a decrease in yield because of hydrocracking reactions which take place in the reforming operation.
The typical feedstocks to catalytic reformers are heavy straight-run (HSR) gasolines and naphthas(82–190°C)] and heavy hydrocracker naphthas. • These are composed of the four major hydrocarbon groups: paraffins, ole- fins, naphthenes, and aromatics (PONA). • Typical feedstocks and reformer products have the following PONA analyses (vol %):
The paraffins and naphthenes undergo two types of reactions in being converted to higher octane components: cyclization and isomerization. • The ease and probability of either of these occurring increases with the number of carbon atoms in the molecules and it is for this reason that only the HSR gasoline is used for reformer feed. • The LSR gasoline [C5-82°C)] is largely composed of lower-molecular-weight paraffins that tend to crack to butane and lighter fractions and it is not economical to process this stream in a catalytic reformer. • Hydrocarbons boiling above 400°F (204°C) are easily hydrocracked and cause an excessive carbon laydown on the catalyst.
REACTIONS Desirable reactions in a catalytic reformer all lead to the formation of aromatics and isoparaffinsas follows: • Paraffinsare isomerized and to some extent converted to naphthenes. The naphthenes are subsequently converted to aromatics. • Olefinsare saturated to form paraffins which then react as in (1). • Naphthenesare converted to aromatics. • Aromatics are left essentially unchanged. Reactions leading to the formation of undesirable products include: • Dealkylationof side chains on naphthenes and aromatics to form butane and lighter paraffins • Cracking of paraffins and naphthenes to form butane and lighter paraffins
As the catalyst ages, it is necessary to change the process operating conditions to maintain the reaction severity and to suppress undesired reactions • There are four major reactions that take place during reforming. • They are: • dehydrogenation of naphthenes to aromatics • dehydrocyclizationof paraffinsto aromatics • Isomerization • hydrocracking
Dehydrogenation Reactions • The dehydrogenation reactions are highly endothermic and cause a decrease in temperature as the reaction progresses. • In addition, the dehydrogenation reactions have the highest reaction rates of the reforming reactions which necessitates the use of the interheaters between catalyst beds to keep the mixture at sufficiently high temperatures for the reactions to proceed at practical rates • The major dehydrogenation reactions are: 1. Dehydrogenation of alkylcyclohexanes to aromatics:
2. Dehydroisomerization of alkylcyclopentanes to aromatics: 3. Dehydrocyclization of paraffins to aromatics:
The dehydrogenation of cyclohexane derivatives is a much faster reaction than either the dehydroisomerization of alkylcyclopentanes or the dehydrocyclization of paraffins, however, all three reactions take place simultaneously and are necessary to obtain the aromatic concentration needed in the reformate product to give the octane improvement needed • Aromatics have a higher liquid density than paraffins or naphthenes with the same number of carbon atoms, so 1 volume of paraffins produces only 0.77 volumes of aromatics, and 1 volume of naphthenes about 0.87 volume. • In addition, conversion to aromatics increases the gasoline end point because the boiling points of aromatics are higher than the boiling points of paraffins and naphthenes with the corresponding number of carbons
The yield of aromatics is increased by: • High temperature (increases reaction rate but adversely affects chemical equilibrium) • Low pressure (shifts chemical equilibrium ‘‘to the right’’) • Low space velocity (promotes approach to equilibrium) • Low hydrogen-to-hydrocarbon mole ratios (shifts chemical equilibrium ‘‘to the right,’’ however, a sufficient hydrogen partial pressure must be maintained to avoid excessive coke formation)
Isomerization Reactions • Isomerization of paraffins and cyclopentanes usually results in a lower octane product than does conversion to aromatics. However, there is a substantial increase over that of the unisomerized materials. These are fairly rapid reactions with small heat effects. 1. Isomerization of normal paraffins to isoparaffins:
2. Isomerization of alkylcyclopentanes to cyclohexanes, plus subsequent conversion to benzene:
Isomerization yield is increased by: 1. High temperature (which increases reaction rate) 2. Low space velocity 3. Low pressure Hydrocracking Reactions The hydrocracking reactions are exothermic and result in the production of lighter liquid and gas products. They are relatively slow reactions and therefore most of the hydrocracking occurs in the last section of the reactor. The major hydrocracking reactions involve the cracking and saturation of paraffins.
Hydrocracking yields are increased by: • High temperature • High pressure • Low space velocity
Low pressure reforming is generally used for aromatics production and the following generalizations hold for feedstocks in the (68–175°C) TBP boiling range: 1. On a mole basis, naphthene conversion to aromatics is about 98% with the number of carbon atoms in the precursor being retained in the product as follows: • Methylcyclopentane produces benzene. • Cyclohexane produces benzene. • Dimethylcyclopentane produces toluene. • Dimethylcyclohexane produces xylene. • Cycloheptane produces toluene. • Methylcycloheptane produces xylene.
2. For paraffins the following number of moles of aromatics are produced from one mole of paraffins having the indicated number of carbon atoms:
FEED PREPARATION • The active material in most catalytic reforming catalysts in platinum. Certain metals, hydrogen sulfide, ammonia, and organic nitrogen and sulfur compounds will deactivate the catalyst • Feed pretreating, in the form of hydrotreating, is usually employed to remove these materials. • The hydrotreater employs a cobalt– molybdenum catalyst to convert organic sulfur and nitrogen compounds to hydro- gen sulfide and ammonia, which then are removed from the system with the unreacted hydrogen. • The metals in the feed are retained by the hydrotreater catalyst. • Hydrogen needed for the hydrotreater is obtained from the catalytic reformer. • If the boiling range of the charge stock must be changed, the feed is redistilled before being charged to the catalytic reformer.
The reformer operating pressure and the hydrogen/feed ratio are compromises among obtaining maximum yields, long operating times between regeneration, and stable operation. • It is usually necessary to operate at pressures from 50 to 350 psig (345–2415 kPa) and at hydrogen charge ratios of 3–8 mol H2/mol feed (2800–7600 scp/bbl). • Liquid hourly space velocities in the area of 1 to 3 are in general use. • The original reforming process is classified as a semiregenerative type because catalyst regeneration is infrequent and runs of 6 to 24 months between regeneration are common. • In the cyclic processes, regeneration is typically performed on a 24- or 48-hour cycle, and a spare reactor is provided so that regenera- tion can be accomplished while the unit is still on-stream. • Because of these extra facilities, the cyclic processes are more expensive but offer the advantages of low pressure operation and higher yields of reformate at the same severity
REFORMING CATALYST • All of the reforming catalyst in general use today contains platinum supported on an alumina base. • In most cases rhenium is combined with platinum to form a more stable catalyst which permits operation at lower pressures. • Platinum is thought to serve as a catalytic site for hydrogenation and dehydrogenation reactions and chlorinated alumina provides an acid site for isomerization, cyclization, and hydrocracking reactions
Reforming catalyst activity is a function of surface area, pore volume, and active platinum and chlorine content. • Catalyst activity is reduced during operation by coke deposition and chloride loss. • In a high pressure process, up to 200 barrels of charge can be processed per pound of catalyst before regeneration is needed. • The activity of the catalyst can be restored by high temperature oxidation of the carbon followed by chlorination. • This type of process is referred to as semiregenerative and is able to operate for 6- to 24-month periods between regenerations. • The activity of the catalyst decreases during the on-stream period and the reaction temperature is increased as the catalyst ages to maintain the desired operating severity. • Normally the catalyst can be regenerated in situ at least three times before it has to be replaced and returned to the manufacturer for reclamation.
Catalyst for fixed-bed reactors is extruded into cylinders 1/32 to 1/16 in. (0.8 to 1.6 mm) diameter with lengths about 3/16 in. (5 mm) • The catalyst for continuous units is spherical with diameters approximately 1/32 to 1/16 in. (0.8 to 1.6 mm).
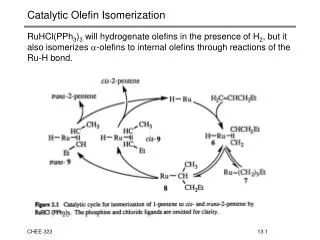
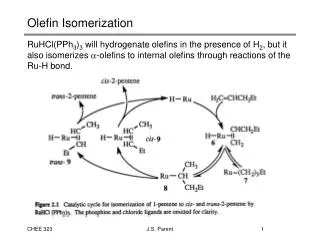
ISOMERIZATION • The octane numbers of the LSR naphtha [C5-180°F(C5-82°C)] can be improved by the use of an isomerization process to convert normal paraffins to their isomers. • This results in significant octane increases as n-pentane has an unleaded (clear) RON of 61.7 and isopentane has a rating of 92.3. • In once-through isomerizationwhere the normal and iso compounds come essentially to thermodynamic equilibrium, the unleaded RON of LSR naphtha can be increased from 70 to about 82–84. • If the normal components are recycled, the resulting research octane numbers will be about 87–93 RONC.
Reaction temperatures of about 200–400°F (95–205°C) are preferred to higher temperatures because the equilibrium conversion to isomers is enhanced at the lower temperatures. • At these relatively low temperatures a very active catalyst is necessary to provide a reasonable reaction rate. • The available catalysts used for isomerization contain platinum on various bases. • Some types of catalysts require the continuous addition of very small amounts of organic chlorides to maintain high catalyst activities • This is converted to hydrogen chloride in the reactor, and consequently the feed to these units must be free of water and other oxygen sources in order to avoid catalyst deactivation and potential corrosion problems
A second type of catalyst uses a molecular sieve base and is reported to tolerate feeds saturated with water at ambient temperature. • A third type of catalyst contains platinum supported on a novel metal oxide base. • This catalyst has 150°F (83°C) higher activity than conventional zeoliticisomerizationcatalysts and can be regenerated. • Catalyst life is usually three years or more with all of these catalysts. • An atmosphere of hydrogen is used to minimize carbon deposits on the catalyst but hydrogen consumption is negligible
The composition of the reactor products can closely approach chemical equilibrium. • The actual product distribution is dependent upon the type and age of the catalyst, the space velocity, and the reactor temperature. • The pentane fraction of the reactor product is about 75 to 80 wt% iso-pentane, and the hexane fraction is about 86 to 90 wt% hexane isomers