Understanding Chemical Bonding: Ionic, Covalent, and Metallic Bonds
Dive into the world of covalent, ionic, and metallic bonds, learn about sharing electrons, electronegativity, naming compounds, and more. Explore different bond types and their characteristics with clear explanations and examples.


Understanding Chemical Bonding: Ionic, Covalent, and Metallic Bonds
E N D
Presentation Transcript
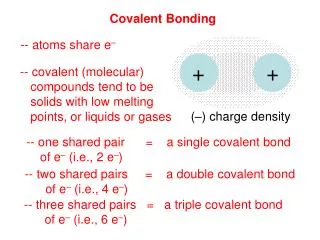
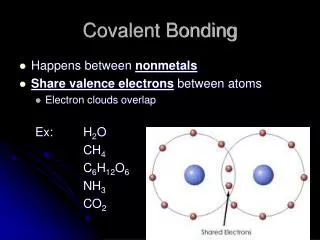
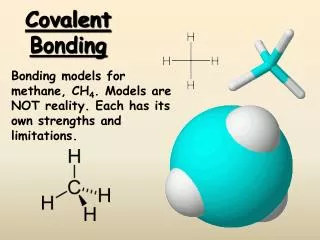
Covalent Bonding Sharing is caring
Types of Bonds Review Transfer Electrons Unit Cell Ionic Molecule Covalent Metallic Share Electrons Delocalized Electrons (sea of e-)
Electronegativity and bond types • Another way to usually tell whether a bond is ionic or covalent is with electronegativities • Electronegativity (Page 194)– the tendency of an atom to gain electrons in a chemical bond. • If the difference between the electronegativities is large, the bond tends to be ionic. If it is small, it tends to be covalent. • Compounds can have both, but usually show one type more than the other chemically, usually ionic).
Types of Covalent Compounds • Diatomic molecules (twins): ________________________________ ________________________________ • NEVER FOUND AS SINGLE ATOMS if “alone” • Their names are just the name of the element H2, N2, O2, F2, Cl2, Br2, I2 There are seven diatomic molecules
Binary Molecular Compounds Non-metals with no H+ ___________________________(no metals or ions) i.e. Does not start with an H. • Steps to name compounds • _________ _________ - use entire name • _________ _________ - change ending to –ide • Prefixes used to indicate _________ of atoms of each p. 832 • Only time that no prefix is used is if the first is a one • Examples: CCl4 – As2O3 – Name first Name second number carbon tetra chloride di arsenic tri oxide
The Prefixes Mono- monocle Di- Dialogue Tri- tricycle Tetra- Tetris Octa- octopus Penta- pentagon
Acids • Always begin with ______ • Two Types: _________ and ___________ H+ binary oxyacid Remember: The number of hydrogen atoms in the acid equals the charge of the anion
Binary Acids H+ O • Binary Acids - ______ but no _______ • Steps to name compounds • Use prefix - _________ • Root of second element plus suffix - _________ followed by the word acid • Example: HCl - hydro -ic hydro chloric acid
Oxyacids O polyatomic ion • – has a ________________, so most have ___ Steps to name compounds • Identify polyatomic ion p. 178 • Use root of anion, change suffix ending and word acid • Suffix rules: ends in ate change to _________ • ends in ite change to _________ Example: HNO3 - -ic -ous nitric acid
Hydrocarbons • a compound containing carbon and hydrogen • Named using prefixes that correspond to the number of carbons in the compound. • 1 – meth • 2 – eth • 3 – prop • 4 – but • After this it used the “normal” prefixes
Hydrocarbons • Ending refers to the type of functional group found in the compound. • The simplest hydrocarbon ends in -ane which means that the hydrocarbon only has single bonds and the number of hydrogen atoms equal 2(Carbon atoms)+2.
Practice Phosphorous pentachloride Binary Molecular Hydroiodic acid Binary Acid Nitrogen Diatomic Hydrocarbon ethane Oxyacid Phosphic acid Chlorous acid Oxyacid Dinitrogen tetrahydride Binary Molecular