Solubility
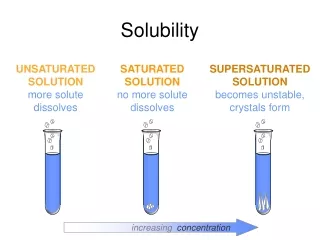
Solubility Crystallize Solute + Solvent Solution Dissolve Unsaturated Solution – Additional solute will still dissolve Saturated Solution – No additional solute will dissolve, equilibrium between solution and undissolved solute

Solubility
E N D
Presentation Transcript
Solubility Crystallize Solute + Solvent Solution Dissolve • Unsaturated Solution – Additional solute will still dissolve • Saturated Solution – No additional solute will dissolve, equilibrium between solution and undissolved solute • Supersaturated Solution – Beyond the point of saturation
Salt Solubility and Health • Sodium urate is formed from uric acid, a natural chemical in the body. Uric acid can also come from foods. • Uric acid in normal amounts remains dissolved in the blood, easily passes through the kidneys and leaves the body as waste. Uric acid in high amounts, however, makes a person more likely to develop gout. • Uric acid crystals deposit in the joints • The amount of uric acid in your blood can change depending on: • What you eat: red meats and internal organs (such as liver and kidneys), some shellfish and anchovies • Your overall health • How much alcohol you drink • What medicines you are taking • Sudden illnesses • The kidneys' ability to rid the body of uric acid (heredity).
Solubility Crystallize Solute + Solvent Solution Dissolve • Factors Affecting Solubility • Energy Requirements • Solvent-Solute interactions (like dissolves like) • Temperature Effects • Pressure Effects (Henry’s Law)
The Solution Process Energy Changes and Solution Formation
The Solution Process Energy Changes and Solution Formation CaCl2 example NH4Cl example
The Solution Process Energy Changes and Solution Formation • Breaking attractive intermolecular forces is always endothermic. • Forming attractive intermolecular forces is always exothermic. • To determine whether Hsoln is positive or negative, we consider the strengths of all solute-solute and solute-solvent interactions: • H1 and H2 are both positive. • H3 is always negative. • It is possible to have either H3 > (H1 + H2) or H3 < (H1 + H2).
The Solution Process Energy Changes and Solution Formation • “Rule”: polar solvents dissolve polar solutes. Non-polar solvents dissolve non-polar solutes. Why? • If Hsoln is too endothermic a solution will not form. • NaCl in gasoline: the ion-dipole forces are very weak because gasoline is non-polar. Therefore, the ion-dipole forces do not compensate for the separation of ions (loss of ion-ion interactions). • Water in octane: water has strong H-bonds. There are no attractive forces between water and octane to compensate for the lost H-bonds when water molecules separate.
Salt Solubility Examples • Some salts have very low solubility in water and reach saturation right away These salts are often referred to as “Insoluble” salts Ion-ion interaction are too strong to be separated by water
Temperature Affects on Solubility • Most solids increase solubility in a liquid with increasing temperature • Most gases decrease solubility in a liquid with increasing temperature
Pressure Affects on Solubility • Solubility of liquids and solids not greatly affected by pressure • Solubility of gases greatly affected by pressure (Henry’s Law) • Increase pressure = Increase solubility • Coca-cola example
Bends and Scuba Diving“Henry’s Law in Action” • Pressure on the body increases at the divers goes deeper • Increased pressure = increased gas in blood • Air is mostly nitrogen so more nitrogen than normal dissolved in our blood • As the diver ascends, the gases come out (pressure decreases) • Fast ascends = lots of bubbles (BAD for our organs and joints!)
Altitude Illness- Mountain Sickness (Form of Hypoxia) • Most likely to be affected are mountain climbers, pilots and persons living at high altitudes above 8,000 feet. • High altitude = low pressure = low oxygen concentration dissolved in blood • In a healthy person at sea level, blood is 95% saturated with oxygen. At 18,000 feet it is only 71% saturated.
Ways of Expressing Concentration • All methods involve quantifying amount of solute per amount of solvent (or solution). • Generally amounts are measures are masses, moles or liters. • Qualitatively solutions are dilute or concentrated. • Definitions:
Ways of Expressing Concentration • Parts per million (ppm) can be expressed as 1 mg of solute per kilogram of solution. • Parts per billion (ppb) are 1 g of solute per kilogram of solution. • Proof by conversion
Ways of Expressing Concentration Mole Fraction, Molarity, and Molality
Ways of Expressing Concentration Mole Fraction, Molarity, and Molality • Converting between molarity (M) and molality (m) requires density of solution • L of solutions to kg of solution • kg of solution = kg solvent + kg solute moles of solute kg of solvent molality =
Colligative Properties • Colligative properties depend on quantity of solute molecules. • Freezing point depression • Boiling point elevation
Lowering the Vapor Pressure • Non-volatile solvents reduce the ability of the surface solvent molecules to escape the liquid. • Therefore, vapor pressure is lowered. • The amount of vapor pressure lowering depends on the amount of solute.
Boiling-Point Elevation • Non-volatile solute lowers the vapor pressure. • Higher Temperature needed to achieve 1 atm of vapor pressure Freezing-Point Depression • Non-volatile solute lowers the vapor pressure, and shift triple point to lower temperature • Lower temperature of triple point produces lower phase change temperature
Colligative Properties Boiling-Point Elevation • At 1 atm (normal boiling point of pure liquid) there is a lower vapor pressure of the solution. Therefore, a higher temperature is required to reach a vapor pressure of 1 atm for the solution (Tb). • Molal boiling-point-elevation constant, Kb, expresses how much Tb changes with molality of particles, m:
Colligative Properties Freezing-Point Depression • The solution freezes at a lower temperature (Tf) than the pure solvent. • Decrease in freezing point (Tf) is directly proportional to molality of particles (Kfis the molal freezing-point-depression constant):