Understanding Atomic Mass and Formula Mass in Chemistry
In chemistry, masses are essential for describing substances. Atomic mass indicates the weighted average of isotopes' masses of an element, measured in atomic mass units (amu). To determine a compound's mass, we calculate its formula mass, which sums the atomic masses of all its constituent atoms. For example, the formula mass of water (H₂O) is 18 amu, derived from its atomic components. This guide also includes practice problems for compounds like NaCl and H₂SO₄ to reinforce understanding.

Understanding Atomic Mass and Formula Mass in Chemistry
E N D
Presentation Transcript
Many measurements are used to describe substances in Chemistry. Since the atoms of different elements have different masses, one of the measurements used is the atomic mass of an element.
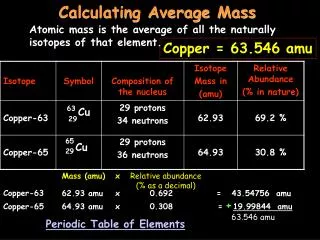
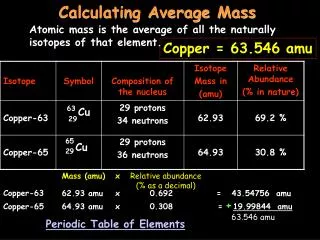
What is atomic mass? ATOMIC MASS weighted average of the masses of the existing isotopes of an element ATOMIC MASS UNIT (amu) a special unit used to describe the mass of an atom of an element
Reinforcement Q: What is the amu of Be? A: Q: What is the amu of Kr? A:
What is formula mass? To find the mass of an entire molecule of a compound, calculate the formula mass of the compound. FORMULA MASS the sum of the atomic masses of all the atoms in a compound
EXAMPLE: a molecule of water (H2O) H : 2 atoms x 1 amu = 2 amu O : 1 atom x 16 amu = + 16 amu Formula Mass 18 amu
Reinforcement Q: What is the formula mass of SO2? A: Q: What is the formula mass of Mg(OH)2? A:
PRACTICE PROBLEMS 1. NaCl