Chemical Reaction Notes
100 likes | 138 Vues
Explore how matter can be transformed through physical and chemical changes. Learn about the processes involved, signs of chemical reactions, and common examples. Discover the difference between physical and chemical changes in matter.

Chemical Reaction Notes
E N D
Presentation Transcript
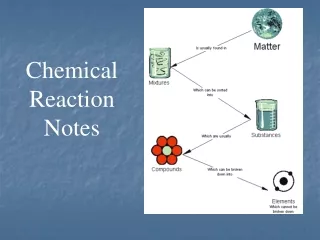
Matter is everything around you. Matter is anything made of atoms and molecules. Matter is anything that has a mass.
Matter can be changed. It can go through physical change or chemical change.
Physical changes are a change in state or form that does not result in a new substance being created.
Some common examples of physical changes are: • melting • freezing • condensing • breaking • crushing • cutting • bending
Chemical changes result in the formation of a new substance via a chemical reaction. • Energy is required for a chemical change to take place.
Chemical Reaction • Process in which one or more substance changes to produce one or more new substances with different chemical and physical properties. • During a chemical reaction, molecular bonds are broken and new bonds are made.
Signs that a chemical change has occurred: • production of a gas (bubbles) • formation of a new odor • production of a precipitate (solid) • change of color • Energy change given off as light, thermal, or electrical energy
Common examples of chemical changes that you may be somewhat familiar with are: • digestion • respiration • photosynthesis • burning • decomposition
Chemical and Physical Changes - Introduction Chemical and Physical Changes