Diffusion and Osmosis
Diffusion and Osmosis. 1. 4. 2. 3. Outline. Learn the concepts of: Diffusion, osmosis, semi-permeable membrane, isotonic, hypertonic, & hypotonic Explore diffusion in a colloid Relationship between diffusion & size Explore diffusion & osmosis in a dialysis bag Dialysis bag experiment

Diffusion and Osmosis
E N D
Presentation Transcript
1 4 2 3 Outline • Learn the concepts of: • Diffusion, osmosis, semi-permeable membrane, isotonic, hypertonic, & hypotonic • Explore diffusion in a colloid • Relationship between diffusion & size • Explore diffusion & osmosis in a dialysis bag • Dialysis bag experiment • Semi-permeable membrane • Water, glucose, starch • Plant/animal cells exposure to water • Hypertonic Env. • Hypotonic Env. • Isotonic Env.
1 4 2 3 Diffusion • Solute molecules moving from an area of high concentration to an area of low concentration • Random motion drives diffusion • Movement is based on kinetic energy (speed), charge, and mass of molecules • Equilibrium is reached when there is an even distribution of solute molecules (water)
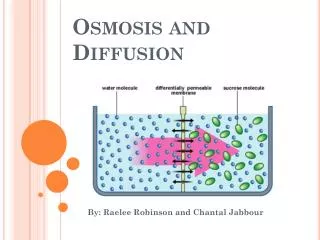
Osmosis • Diffusion of water through a semi-permeable membrane • Semi-permeable: permeable to solvents (WATER), but not to large molecules • High [water] to low [water] • Dissolved molecules (i.e. glucose, starch) are called solutes • REMEMBER: Water = solvent Glucose, Starch = solutes
2 1 1 3 6 4 5 Diffusion Rates of Various Dyes • How do different MW solutions migrate through a colloid? • Colloid – suspension of large, insoluble particles in a fluid medium • Agar plate • Measure the rate of diffusion of various dye (differ in MW) • Measure diameter at T90 (minutes) • 3 known(s) (crystal violet, malachite green, potassium permanganate) • 3 unknowns Part 1 pg. 82
Diffusion Rates of Various Dyes • How do different MW solutions migrate through a colloid? • Standard Curve • Y-axis: molecular weight (MW) • X-axis: distance migrated (mm) ? • Inverse Relationship • Higher MW, slower diffusion • Lower MW, faster diffusion
Dialysis bag Glucose Iodine Starch T0 T80 Weigh Weigh Dialysis Bag Experiment • Dialysis Bag • Semi-permeable membrane • Water, glucose, & starch • What passes through the dialysis bag? • Glucose (Benedict’s Test) • Starch (Iodine Solution) • Time Course Experiment • Every 10 minutes, measure the amount of glucose & starch present in the beaker Part 2 pg. 83
Dialysis Bag Experiment - Results Lab Manual, page 84
Dialysis Bag Experiment - Controls • Negative control for dialysis bag experiment (performed by TA) • Instead of using dialysis tubing, I will be using a plastic bag. • I will determine the weight, Benedict‘s result, & iodine results at time 0 and 80 minutes Biochemical test controls (in pairs) • set up test control of iodine test w/ glucose and starch • set up test control of Benedict‘s test w/ glucose and starch
Effect of Water on Cells • Hypertonic Environment • High [solute], low [water] • Hypotonic Environment • High [water], low [solute] • Isotonic Environment • [water] = [solute] Hypertonic Hypotonic Isotonic Part 3 pg. 85
Cellulose in cell wall Osmosis in Living Cells
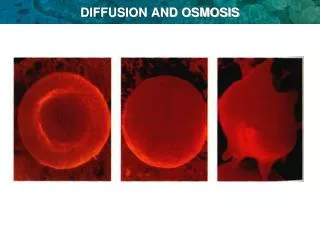
Isotonic Predictions? Hypertonic Hypotonic Osmosis in Red Blood Cells • Observe sheep RBCs via a wet mount of the sample • Aliquot one drop the following solutions with a ½ drop of RBC to a slide • 0.9% saline • 10% NaCl • Distilled water • Record observation in the table on page 85 Crenation
Hypertonic Predictions? Hypotonic Osmosis in Plant Cells Plasmolysis • Observe Elodea leaves via a wet mount of the sample • Aliquot two drops the following solutions with a new Elodea leaf to a slide. Incubate for 10 minutes @ room temp. • 10% NaCl • Distilled water • Record observation in the table on page 85
Plan of attack . . . • Set-up Part I – Molecular Weight Diffusion in Colloid • Incubate for 90 minutes • Set up Part II – Dialysis Bag • Time course: 80 minutes (time-point every 10 minutes) • Part III – Osmosis in Living Cells
Animations Dialysis Bag Experiment • http://ccollege.hccs.cc.tx.us/instru/Biology/AllStudyPages/Diffusion_Osmosis/Baggif.swf Elodea Cell • http://ccollege.hccs.cc.tx.us/instru/Biology/AllStudyPages/Diffusion_Osmosis/Elodeagif.swf Osmosis • http://ull.chemistry.uakron.edu/genobc/animations/osmosis.mov
Transportation of Molecules • Passive Transport • Movement of molecules across a semi-permeable membrane • no energy required • Active Transport • Movement of molecules across a semi-permeable membrane against a concentration gradient with a protein • ENERGY – ATP • Facilitated Diffusion • Movement of molecules across a semi-permeable membrane with a protein • no energy required