Mastering Organohalides: Substitution and Elimination Reactions
590 likes | 608 Vues
Explore the synthesis and properties of alkyl halides, from Grignard reagents to radical halogenation. Dive into SN2 and SN1 reactions, mechanisms, and influencing factors for reaction rates.

Mastering Organohalides: Substitution and Elimination Reactions
E N D
Presentation Transcript
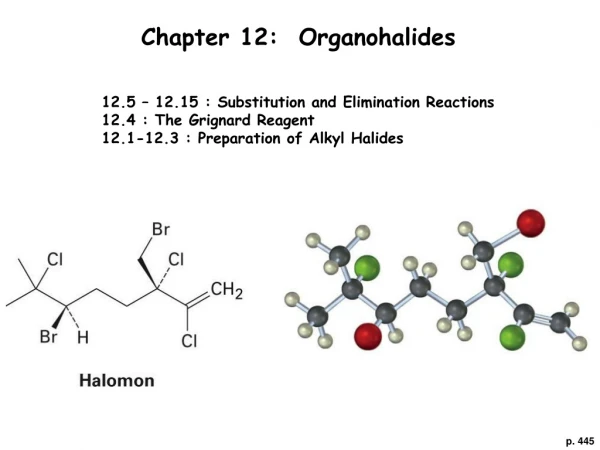
Chapter 12: Organohalides 12.5 – 12.15 : Substitution and Elimination Reactions 12.4 : The Grignard Reagent 12.1-12.3 : Preparation of Alkyl Halides p. 445
Properties of Alkyl Halides Table 12-1, p. 446
Properties of Alkyl Halides p. 447
Synthesis of alkyl halides : review • Addition of HX to an alkene (section 7.6) p. 227
Synthesis of alkyl halides : review • Addition of HX to an alkene (section 7.6) • Obeys Markovnikov’s Rule (section 7.7) • Carbocations that can rearrange, will rearrange (section 7.10) p. 239
Synthesis of alkyl halides : review 2. Addition of X2 to an alkene (section 8.2)
Synthesis of alkyl halides : review 3. Radical Halogenation (section 6.3)
Synthesis of alkyl halides : New • From alkenes • Allylicbromination with N-BromoSuccinimide • From Alcohols • Using strong acid • Via Tosylates • Using SOCl2 and PBr3
Synthesis of alkyl halides : New • From alkynes • Allylicbromination with N-BromoSuccinimide • From Alcohols • Using strong acid • Via Tosylates • Using SOCl2 and PBr3
Synthesis of alkyl halides : New • From alkynes • Allylicbromination with N-BromoSuccinimide • From Alcohols • Using strong acid • Via Tosylates • Using SOCl2 and PBr3
Synthesis of alkyl halides : New • From alkynes • Allylicbromination with N-BromoSuccinimide • From Alcohols • Using strong acid • Via Tosylates • Using SOCl2 and PBr3
The Suzuzi – Miyaura Cross Coupling The Nobel Prize in Chemistry 2010 "for palladium-catalyzed cross couplings in organic synthesis" Richard F. Heck Ei-ichiNegishi Akira Suzuki
Characteristics of the SN2 reaction • Rate equation
Characteristics of the SN2 reaction • Rate equation • Mechanism
The Walden Inversion Cycle (1896) (-)-Maleic Acid [a]D = -2.3 (+)-chlorosuccinic acid (+)-Maleic Acid [a]D = +2.3 (-)-chlorosuccinic acid Fig. 12-2, p. 455
Characteristics of the SN2 reaction • Rate equation • Mechanism • Molecular factors effecting SN2 reaction rate
The Effect of Reactant and Transition Structure Energies on Reaction Rate Fig. 12-6, p. 460
Effect of ELECTROPHILIC CARBON on SN2 rate Fig. 12-7, p. 461
Effect of ELECTROPHILIC CARBON on SN2 rate
Effect of NUCLEOPHILE on SN2 rate Table 12-2, p. 462
Characteristics of the SN1 reaction • Rate equation
Characteristics of the SN1 reaction • Rate equation • 2. Mechanism of SN1 reaction
Characteristics of the SN1 reaction • Rate equation • 2. Mechanism of SN1 reaction • 3. Molecular factors effecting SN1 reaction rate
Effect of electrophilic carbon on SN1 rate:Carbocation Stability
Effect of electrophilic carbon on SN1 rate: Resonance in Carbocations Fig. 12-13, p. 471
Bond Disassociation Energies and Carbocation Stability p. 472
Bond Disassociation Energies and Carbocation Stability < p. 472
Effect of LEAVING GROUP on SN1 rate Effect of LEAVING GROUP on SN2 rate p. 472
Effect of Solvent on SN1 rate Fig. 12-15, p. 474