Diffusion and Osmosis
This overview focuses on the principles of diffusion and osmosis, exploring molecular motion and the kinetic energy of molecules. It introduces Brownian movement, Fick's First Law of Diffusion, and the role of semi-permeable membranes in cellular processes. The concepts of osmolarity and tonicity are examined, differentiating between hypertonic, hypotonic, and isotonic solutions, and their effects on cells. Additionally, the text discusses factors affecting the rate of diffusion and dialysis, as well as water uptake by plants, emphasizing the importance of adhesion, cohesion, and water potential.

Diffusion and Osmosis
E N D
Presentation Transcript
Diffusion and Osmosis Bio 21L Spring 2003
Molecular Motion • Molecules posses kinetic energy. • Random motion • Rotational, translation & vibration movements • Brownian Movement: random, zigzag movement, arising due to collisions of molecules with particles. • BM is typical of colloids.
Diffusion • Net movement of molecules from an area of high concentration to one of lower concentration • Fick’s First law of Diffusion: Rate of diffusion is directly proportional to concentration
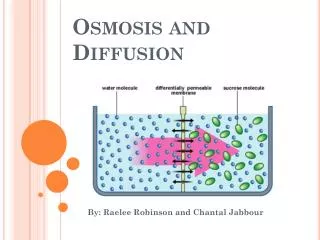
Osmosis • Diffusion of molecules across a semi-permeable membrane from a place of higher concentration to one of lower concentration until the concentration on both sides is equal • Osmolarity: Concentration of osmotically active particles • Tonicity: The osmotic pressure/tension of a solution wrt blood • Hypertonic solutions: are @higher osmotic pressure than the reference solution
Hypertonic solutions: are @ higher osmotic pressure than the reference solution. Exosmosis • Hypotonic: are @ lower pressure than reference. Endosmosis • Isotonic: are @ the same osmotic pressure as reference; e.g. 0.9% NaCl Sol; 5% sucrose sol with human blood cells
Dialysis • Separation of molecules based on size, using a semi-permeable membrane. • Semi-permeable membranes: natural-cell membranes; synthetic- nitrocellulose memb. • Factors affecting rate: Pore size of memb; Size of molecule; polarity of molecules; tonicity of solution outside dialysis bag
Water Potential ۴ & Uptake of Water by Plants • Forces of adhesion & cohesion • Evaporation & transpiration at leaf tip • Generation of water potential • Endosmosis into root cells • Water uptake
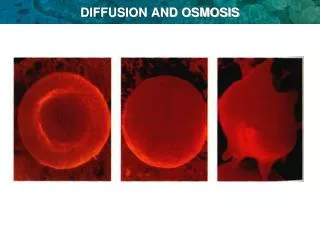
Haemeolysis & Crenation: Ghost Studies • Loss of water by cells- cell shrinkage or crenation • In RBCs-leaves just membranes-used extensively to study memb structure • In plant cells- cell shrinkage- allows plant cell memb to be seen • Plasmolysis-rupturing plant cells by dunking in hypotonic solution. • Turgid cell- plant cells that maintain a good osmolarity & tonicity- appear round & happy!