Condensation Reactions
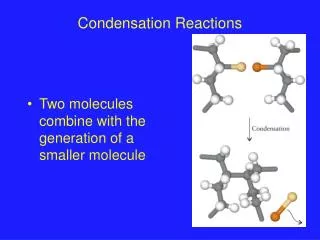
Condensation Reactions. Two molecules combine with the generation of a smaller molecule. Condensation Reactions. Reaction of Acetic Acid and Ethanol. Looking at the Reaction Mechanism. The carbonyl carbon is: Electron deficient In a trigonal planar geometry 120º between substituents

Condensation Reactions
E N D
Presentation Transcript
Condensation Reactions • Two molecules combine with the generation of a smaller molecule
Condensation Reactions • Reaction of Acetic Acid and Ethanol
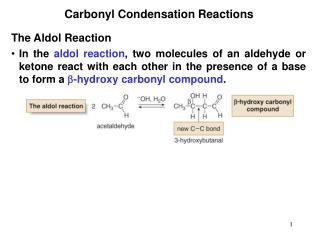
Looking at the Reaction Mechanism • The carbonyl carbon is: • Electron deficient • In a trigonal planar geometry • 120º between substituents • The carbonyl oxygen is pulling electrons towards it • Resonance stabilization • The Lone Pair of the alcohol oxygen can react with the carbonyl carbon to set the whole thing in motion • Remember your VSEPR Geometry
Condensation Reactions: Making Lipids from Sugars and Fatty Acids • Your cells can synthesize lipids from glycerol and fatty acids in a condensation reaction
Condensation Reactions: Forming a Peptide Bond What are the amino acids in the figure? What function group is formed? Its not really this simple, but it illustrates a point!
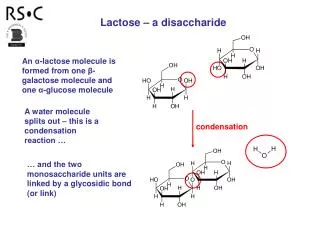
Hydrolysis: The Opposite of Condensation • In a hydrolytic reaction, we add the elements of water (H+ and OH-) across a bond • Many enzymes use this kind of reaction to degrade polymers • Lipases: Hydrolyze lipid esters • Glycosidases: Hydrolyze carbohydrate polymers • Peptidases: Hydrolyze peptide bonds • Compound Name + ase : Usually indicates a hydrolase (but not always!) • If it isn’t a compound name and ase, then it usually does something else: • Lyase • Reductase • Kinase • Transferase
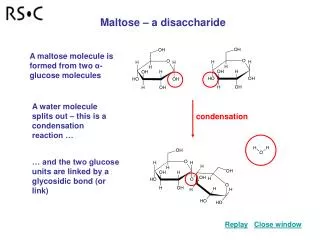
Hydrolysis of Sugar Polymers • We add water across the Glycosidic Bond of Maltose to break it and generate 2 monomers • Catalyzed by a glycosidase (Maltase perhaps?)
Hydrolysis of Peptides • Dipeptide (What are the amino acids) is hydrolyzed to ??? • Catalyzed by a peptidase or a protease
Amino Acids • Amino acids are the building blocks of proteins • They consist of an amino group bonded to an -carbon, a hydrogen bonded to the -carbon and a carboxylic acid
Amino Acids and Stereochemistry • The -carbon is all amino acids except for glycine is chiral • Stereoisomers exist that is non-superimposable • Any carbon with 4 different substituents can be chiral • We describe the chirality of the -carbon as being Levorotary or Dextrorotary • L- or D- • Refers to how the molecule rotates polarized light
Amino Acid Side Chains: Where the Action is! • The amino acids are classified according to the chemical character of the R-grop attached to the -carbon • Important Criteria: • Polar or Nonpolar side chains • Acidic or Basic • Charged or uncharged Polar residues
Side Chain Classification • Nonpolar (hydrophobic) Amino Acids G, A, V, L, I, P, F, W, M • These amino acids have aliphatic side chains • Phenylalanine and Tryptophan are aromatic • Proline is cyclic • Induces turns in proteins
Side Chain Classification • Polar, Uncharged Amino Acids S, T, Y, C, N, Q • S, T, Y have hydroxyl groups (-OH) • C has a sulfhydryl (-SH) • N and Q have amide side chains • Uncharged at neutral pH
Side Chain Classification • Acidic Amino Acids D and E have carboxylic acids on their side chains • The side chains are negatively charged at neutral pH • This means the pKa’s of the side chains are less than 7
Side Chain Classification • Basic Amino Acids H, K and R have side chains that are positively charged at neutral pH • Because these side chains have basic groups, they accept protons at pH values lower than the pKa of the side chain
Titrating Amino Acids • Free amino acids can have up to 3 pKa values associated with them • Carboxylic acid • Amino group • R-group • The carboxylic acid group has the lowest pKa (~2.0) • The pKa of the -amino is around 9-10 • D, E, H, C, Y, K and R have R-groups that can ionize and their pKa’s range from ~4 to 12
Titrating and Amino Acid: Alanine • We’ll start at a pH of 1 • The carboxylic acid and the amino group are protonated • As we start adding base, more and more of the carboxylic acids start losing protons until we reach pH 2.34 (the pKa of COOH) • At this concentration, [NH3+CHCH3COOH]=[NH3+CHCH3COO-] (same as we learned with regular titrations) • As we add more base, we deprotonate all the carboxylic acids • Midway up the sharp slope increase • For alanine, this is the isoelectric point • As we add more base, we’ll start deprotonating the -amino group until we reach pH=9.69 (the pKa of the group) • [NH3+CHCH3COO-]=[NH2CHCH3COO-] • Finally we can keep adding base until the only species is: NH2CHCH3COO-
Titrating and Amino Acid: Histidine • We’ll start at a pH of 1, the only species is the fully protonated form. • pK1 (COOH) = 1.82 • pK2 (Imidazole nitrogen) = 6.0 • pK3 (Amino) = 9.17 • As we start adding base, the pH increases as the carboxylic acid converts to carboxylate • At pK1, the concentration of the carboxylate specie equals the concentration of the carboxylic acid species • As we add more base, we start deprotonating the imidazole nitrogen • At pK2, the conc. of the deprotonated imidazole group equals that of the protonated state • The pI is reached then the imidazole group is completely deprotonated • As we add more base, we’ll start deprotonating the -amino group until we reach pH=9.17 (the pKa of the group)
Amino Acid Titrations • At the isoelectric point, the molecule has zero net charge • The pH where this occurs is called the pI • We can calculate the pI of an amino acid using the following equation: • We average the pK values from the higher pKa that lost a hydrogen and the lowest pKa that is still protonated • For example: Histidine • pK1 = 1.82 • pK2 = 6.0 • pK3 = 9.17 • We’d use the last two values • Usually it will be the alpha amino and the R group pK’s that are used But we must take care to use the correct pK values!
The Peptide Bond • Amino acids are joined together in a condensation reaction that forms an amide known as a peptide bond
The Peptide Bond • A peptide bond has planar character due to resonance hybridization of the amide • This planarity is key to the three dimensional structure of proteins