What’s the Matter?
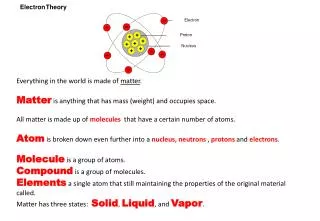
What’s the Matter? . Matter. What’s the Matter? p. 7. Made of Atoms. “Stuff” that Everything is made of!. Can be a solid, liquid, gas, or plasma. Matter. Has Volume= Takes Up Space. Has Mass= The Amount of Matter in an Object. What’s the Matter?. Made of Atoms.

What’s the Matter?
E N D
Presentation Transcript
What’s the Matter? Matter
What’s the Matter? p. 7 Made of Atoms “Stuff” that Everything is made of! Can be a solid, liquid, gas, or plasma Matter Has Volume= Takes Up Space Has Mass= The Amount of Matter in an Object
What’s the Matter? Made of Atoms “Stuff” that Everything is made of! Can be a solid, liquid, gas, or plasma Matter Has Volume= Takes Up Space Has Mass= The Amount of Matter in an Object
States of Matter Brainstorm p. 2 • How many states of matter are there? • What are the states of matter? • Draw what you think the particles look like on a microscopic level • Describe their motion.
SOM Animations • http://www.chemtutor.com/sta.htm • http://www.kentchemistry.com/links/Matter/HeatingCurve.htm
Fahrenheit K Kelvin ˚C Celsius ˚F
Can you freeze air? • Nitrogen (what 78% of the air is made of) freezes at -210 degrees Celsius. Oxygen (air is about 20% oxygen) freezes at -222.65 degrees Celsius. So if you want to have a block of solid air it would have to be at least 222.65 degrees below zero.
Can gold become a gas? • Any element can become a gas; you just need to heat it past its boiling point. For gold, that’s 2,856 degrees Celsius (5,173 degrees Fahrenheit), which is hotter than the temperature in an arc furnace so it’s difficult to just boil a bar of gold. But you can create small amounts of gold gas by bombarding it with a high-energy beam of electrons in a vacuum. This knocks atoms of gold free from the solid mass and they’ll fly around and coat anything in the vacuum chamber. Electron beam vapour deposition is used to create very thin gold coatings for the electronics, medical and space industries. Gold coatings can also be used as a lubricant in machinery. • http://www.howitworksdaily.com/science/can-gold-become-a-gas/
4 States of Matter S.O.M. • Solid • Liquid • Gas • Plasma
Plasma • In plasma TVs, little pockets of gas are excited with electricity disrupting the normal balance of atoms so there are lots of free ions and electrons, turning them into plasma, which creates a light. • If you super cool gas, you get a BEC superatom wave. Warm it up, and the wave will return to a gas. • http://www.sciencelearn.org.nz/Science-Stories/Strange-Liquids/States-of-matter
Dry Ice • What is Dry Ice? • Solid Carbon Dioxide • I thought Carbon Dioxide was a Gas!? • YES, but at a temperature of -109.3 degrees Fahrenheit it is a SOLID! • Why is Mr. Horinger Wearing Gloves? • Because Dry ice is soooooo cold that it can actually burn your skin!
Dry Ice & Halloween • Why is it used at Halloween time? • Because it goes through a cool process known as SUBLIMATION that creates a fog-like effect • What is SUBLIMATION? • Sublimation is when a Solid skips the liquid phase and goes right to a Gas! Solid Gas
General (Physical) Properties • Physical properties may be observed without changing the nature of the matter. • Boiling point is a physical property • When a liquid boils, the vapor may be condensed to get the liquid back • The liquid is still the same substance
What are some Physical Properties? • Properties you can see, such as color, shape, hardness, and texture • Properties that are easily measured, such as mass, volume, density, melting point, boiling point • Note! Freezing and melting point are the same temperature! • Density = Mass ÷ Volume • Is the ability to be attracted to a magnet a physical property?
How do we measure mass and volume? • Mass is measured on a balance or scale. Common units are grams, milligrams, and kilograms • Liquid volume is measured with a graduated cylinder. Common units are liters and milliliters • Solid volumes may be calculated with formulas or by water displacement Common units are cm3 or milliliters.
Let’s Try Some More Density Problems 1) If the mass of a rock is 500g and its volume is 25cm3, what is its density? • If the density of a liquid is 1.2 g/ml, and its volume is 10ml, what is its mass? • If Bob’s mass is 80kg and his density is 1.6kg/l, what is his volume?
Answers: D = M ÷ V • 1) 500g ÷ 25cm3 = 20g/cm3 • 2) 1.2g/ml x 10ml = 12g • 3) 80kg ÷ 1.6kg/l = 50L M D V
WEIGHT • Weight is the pull of gravity on mass. • Weight changes in response to gravitational pull. • A person’s mass will remain the same on the earth or on the moon, but she will only weigh 1/6 as much on the moon.
PHASES OF MATTER • The most common phases of matter are solid, liquid, and gas • Plasma is a high energy phase found in stars • When a substance changes phase, energy (heat) is lost or gained, but temperature remains the same.
The northern lights are caused by plasma streams from the sun hitting our atmosphere. This causes electrons in atmospheric gases to be excited to a higher energy level, And when they drop back down, a photon of light is emitted.
Types of Phase Changes • Solid to liquid: Melting • Liquid to gas: Vaporization • Gas to liquid: Condensation • Liquid to solid: Freezing • Solid to gas: Sublimation
Physical vs. Chemical Properties vs. Changes BrainPOP Property Changes
Physical OR Chemical Properties? Does not change the identity of the matter Observed with your senses Flammability Color Odor Reactivity Mass Volume Conduction of Heat Changes into a new substance with different properties State of Matter – Solid, Liquid, Gas Change of State – Melting, Freezing, Boiling
Physical OR Chemical Properties? Malleability, Ductility, or Solubility Shape Density Not as easy to observe Length Reactivity with water, oxygen, or air Hardness or Luster Heat of combustion Specific Heat Sublimation, Evaporation, or Condensation New substances are formed pH
Physical OR Chemical Changes? Boiling, Melting, Freezing Burning Mixing, Crushing Sanding Cooking Often easy to observe Do not change the substance Producing Heat, Light, or Odor Affects 1 or more properties of a substance. Crushing or Breaking Reacting Color Changes Fizzingor Foaming
Physical OR Chemical Changes? Smashing a pumpkin Chewing gum Digesting a sandwich Wood rotting Smells like coffee and is brown Tastes bitter Dissolves in water Milk souring Iron rusting Crushing a can Mixing cake batter Decomposing an apple
Physical OR Chemical Changes? Breaking glass Cutting a piece of pie Baking Soda & Vinegar Slicing Bread Crumpling paper Boiling water Kool-aid dissolving in water Cracking an egg Baking a cake Burning a marshmallow Melting an ice cube
Chemical Properties and Changes • Chemical properties describe a substances’ ability to change into a different substance. A chemical change is the process by which the substance changes.
Examples of Chemical Properties • Flammability • Ability to support burning • Ability to combine with other chemicals in a chemical reaction.
Examples of Chemical Changes • Rusting • Photosynthesis • Explosions • Burning • Digestion • Baking food • Fermentation
What are indicators of a chemical change? • Gas given off • Heat absorbed or released • Light given off • Color change • Oxidation of a metal • New substances are formed
Physical Change vs. Chemical Change
Physical Change A change in a substance that does not change the identity of the substance.