Magnetic Dipole Moment
1.48k likes | 6.59k Vues
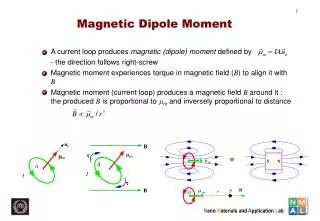
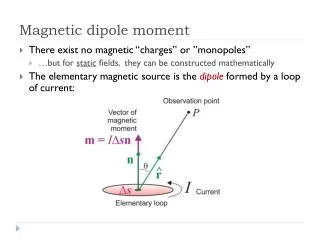
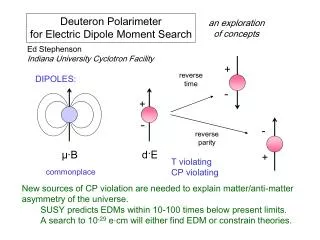
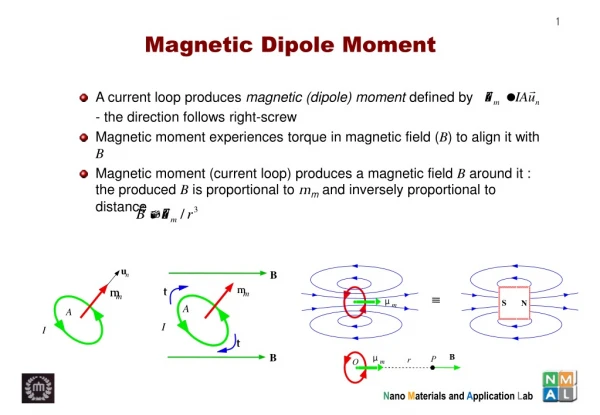
u. n. m. m. A. I. B. m. t. m. µ. S. N. m. A. I. t. B. µ. P. B. r. O. m. Magnetic Dipole Moment. A current loop produces magnetic (dipole) moment defined by - the direction follows right-screw

Magnetic Dipole Moment
E N D
Presentation Transcript
u n m m A I B m t m µ S N m A I t B µ P B r O m Magnetic Dipole Moment • A current loop produces magnetic (dipole) moment defined by - the direction follows right-screw • Magnetic moment experiences torque in magnetic field (B) to align it with B • Magnetic moment (current loop) produces a magnetic field B around it : the produced B is proportional to mm and inversely proportional to distance
Atomic Magnetic Moment • An orbiting electron in atom is a current loop and has a magnetic moment • Since the angular momentum L of the electron is • The ratio e/2m = gyromagnetic ratio • (-) sign indicates morb is in opposite direction of L
z B m µ z s p i n S S z Atomic Magnetic Moment • Electron has an intrinsic angular momentum S (spin) and the spin has spin magnetic moment • The gyromagnetic ratio is a factor of 2 greater • In magnetic field along z-direction, the spin angular momentum is quantized • The spinning electron experiences torque that causes mspin to precess about B • The average magnetic moment mz along the field is • b is called Bohr magneton (9.27x10-24 Am2 or J/T) = magnetic moment of the spin of a single electron along the field
I l B o I I A B M I Magnetization • Magnetic field generated by solenoid is given by • mo is permeability of free space (H/m) • n=number of turns per unit length • Now put a material inside of solenoid, then each atom of the material respond to Bo and develops a magnetic moment mm (the material is magnetized) • Define the magnetic vector M (magnetic dipole moment per unit volume) as to describe the extent of magnetization made by each current loop of atom normal to Bo
S u r f a c e c u r r e n t s S u r f a c e c u r r e n t s Magnetization • In the cross-section of the medium, the current loop cancel each other except at the surface → surface current • Total magnetic moment is M·(volume)=MAl • Suppose the magnetization current on the surface per unit length is Im, then the total magnetic moment is (current)x(cross-sectional area) = • Therefore • Magnetization of a medium = surface current per unit length • Note that magnetization current is not due to a flow of free carriers, but due to localized electronic currents in atoms of the solid
I B M I m l I B Fig. 8.8 : The field in the material inside the solenoid is due to the conduction current I through the wires and the magnetization current Im on the surface of the magnetized m B B M medium, or = + . o o Magnetic Field Intensity • The magnetic field within the medium arises from both current I’ in the solenoid and the magnetization current Im • Bo or B-moM is the contribution of the external current to magnetize the material → define magnetizing field H by • The magnetizing field is also called magnetic field intensity (A/m) • H is the total conduction current per unit length (nI) • H is the cause and B is the effect
Magnetic Permeability/Susceptibility • Magnetic permeability (what extent the material is permeable by the magnetic field?) of a medium is defined by • Relative permeability of a medium is the fractional increase in the magnetic field with respect to the field in free space • Therefore, • The dependence of M on H (similar to the dependence of P on E) is expressed by magnetic susceptibility of the medium • Therefore,
Type c c Comment and examples vs. T m m values) (typical Diamagnetic Negative and T Atoms of the material have closed shells. Organic independe nt small materials, e.g. many polymers; covalent solids, - 6 ( – 10 ) e.g. Si, Ge, diamond; some ionic solids, e.g. alkalihalides, some metals e.g. Cu, Ag, Au. Negative and Below a critical Supercond uctors large ( – 1) temperature Paramagnetic Positive and T Due to the alignment of spins of conduction Independent of small, electrons. ( Alkali and transition metals. - 5 - 4 10 – 10 ) Positive and Curie or Curie - Weiss Materials in which the constituent atoms have a small law permanent magnetic mo ment, e.g. gaseous and - 5 (10 ) c liquid oxygen, ferromagnets (Fe), C T T = / ( – ) m C antiferromagnets (Cr) and ferrimagnets (Fe O ) at 3 4 high temperatures. Ferromagnetic Positive and Ferromagnetic below May possess a large perma nent magnetization very large and paramagnetic even in the absence of an applied field. above the Curie Some transition and rare earth metals, Fe, Co, Ni, temperature Gd, Dy Antiferromagnetic Positive and Antiferromagnetic Mainly salts and oxides of trans ition metals e.g. small below and a MnO, NiO, MnF , and some transition metals, – 2 paramagnetic above Cr, Mn. the Neel temperature Ferrimagnetic Positive and Ferrimagnetic below May possess a large permanent magnetization very large and paramagnetic even in the absence of an applied fi eld. above the Curie Ferrites. temperature Magnetism
2 4 s 6 3 d m 2 l m 1 l L o w e r e n e r g y H i g h e r e n e r g y J e C o N i + F e G d r 0 r d M n C r Ferromagnetism • Fe atom (3d64s2) has 4 unpaired electrons with parallel spins by Hund rule • 4b spin magnetic moment → 2.2b • The electrostatic energy of a paired spin-up and spin-down electrons at the same orbital (ml) is higher due to Coulomb repulsion than the energy of the electrons with parallel spin (same ms) at different orbitals (different ml) → Exchange interaction (Pauli exclusion principle + electrostatic interaction) • Exchange energy for two atoms with outer electrons at a distance r is • S is spin angular momentum • Je is exchange integral • Antiferro : Je <0, S1S2(↑↓)<0→ Eex <0 • Ferro : Je >0, S1S2 (↑↑)>0 → Eex <0 : spin spontaneously align to reduce exchange energy
M T ( ) s a t M ( 0 ) s a t 1 I r o n 0 . 8 0 . 6 0 . 4 0 . 2 0 0 0 . 2 0 . 4 0 . 6 0 . 8 1 T / T C Curie Temperature • Saturation magnetization (Msat) is the max magnetization in ferromagnets when all magnetic moments align • At high temperature, lattice vibration agitates spins and ferromagnetism disappears at Curie temperatureTc • kTc ~ Eex
N M S C l o s u r e d o m a i n 9 0 ° d o m a i n w a l l E a s y d i r e c t i o n N S G r a d u a l r o t a t i o n o r [ 0 0 1 ] z o f m a g n e t i c m o m e n t s N S S N N S o r [ 0 0 1 ] – z D o m a i n A E a s y d i r e c t i o n l B l o c h w a l D o m a i n B A g r a i n w i t h d o m a i n s Magnetic Domain • Fe is not magnetic in general, why? → magnetic domain : a region of crystal in which all the spin magnetic moment is aligned • Magnetostatic energy = potential energy stored in a magnetic field • One domain has external magnetic field • Domain structure can remove external field or reduce magnetostatic energy • Domain wall (Bloch wall) is created • Domain has preferred direction for magnetization : easy direction
[ 1 0 0 ] H A A B B ´ 4 - 1 H M a g n e t i z i n g f i e l d ( 1 0 A m ) 0 1 2 3 4 2 M [ 1 0 0 ] s a t ) 1 [ 1 1 0 ] - m 1 . 5 A [ 1 1 1 ] 6 0 H a r d 1 ´ [ 1 1 1 ] 1 P ( D n M e d i u m o i t a [ 1 1 0 ] z i C t e n 0 . 5 g a M E a s y B [ 1 0 0 ] A O 0 0 0 . 0 1 0 . 0 2 0 . 0 3 0 . 0 4 0 . 0 5 0 . 0 6 m H A p p l i e d m a g n e t i c f i e l d ( T ) o Magnetic Anisotropy • Magnetization occurs by domain wall migration to enlarge the volume having specific spin direction • Magnetic anisotropy : difference in magnetic properties with crystallographic direction • Easy direction • Hard direction • Fe • [100] easy axis • [111] hard axis • Magnetocrystalline anisotropy energy (K) : the excess energy required to magnetize in particular direction with respect to the easy direction
H H H H M d M M M s a t c e a b c d M r R e v e r s i b l e I r r e v e r s i b M R o t a t i o n o f M S a t u r a t i o n o f e b o u n d a r y b o u n d a r y m o t i o n m o t i o n H b f a H H O H c x x + Magnetization Process Remanent or residual magnetization Coercivity or coercive field Barkhausen effect : jumps in magnetization due to jerking in domain wall movement induced by defects
B d B s a t B r H H B r B s a t g B B H a r d S o f t – H H – B M-H • M-H curve shows hysteresis • M-H can be converted to B-H • B=moM+moH • The area of hysteresis is the energy dissipated per unit volume per cycle of the applied field • Soft magnetic material : easy to magnetize • Requires low magnetic field intensity in magnetization and demagnetization • Small hysteresis loop area (low loss) → motor, transformer, inductor • Hard magnetic material • Large Hc → Magnetic storage
B B B o f f S u p e r c o n d u c t o r I P e r f e c t c o n d u c t o r I T T > T T T T < < c c c Superconductivity • Superconductivity : no resistance to current flow below a critical temperature Tc • High Tc superconductor • La-Ba-Cu-O, 35K • Y-Ba-Cu-O, 95K • Hg-Ba-Ca-Cu-O, 130K • Meissner effect : perfect diamagnetic behavior of the superconductors or expel of magnetic field from the bulk • Develop M opposing to B (perfect diamagnetism, cm= -1) • In perfect conductor, no field rejection occurs • For B =0, it disappears in SC, but the decreasing field induce current in the perfect conductor