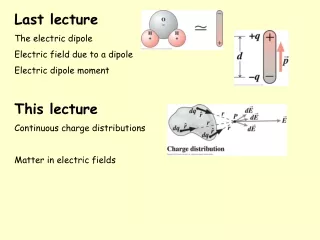
DIPOLE MOMENT
DIPOLE MOMENT. Electronegativity Polarity of Bonds Dipole Moment Dipole Moment of Polar Covalent Bond Dipole Moment of Non Polar Covalent Bond Example of Dipole Moment. Next. ELECTRONEGATIVITY.

DIPOLE MOMENT
E N D
Presentation Transcript
DIPOLE MOMENT Electronegativity Polarity of Bonds Dipole Moment Dipole Moment of Polar Covalent Bond Dipole Moment of Non Polar Covalent Bond Example of Dipole Moment Next
ELECTRONEGATIVITY Definition: Electronegativity is chemical property that describes the ability of an atom to attract electron towards itself in a covalent bond. Symbol: “X” Trends In Electronegativity: Electronegativity increases across the period. Decrease down the group. Fluorine is most electronegative element. Cesium is least electronegative element. Exceptions: Gallium and Germanium have higher electronegative than Aluminum and Silicon respectively because of the D-block contradiction. Order In Decreasing Electronegativity: F>O>N>Br>I>P>H>Si
BondPolarity Definition: When there is a bond between atom with (difference< 1.7) different electronegativity , then more electronegative atoms has a partial negative charge, and other has a partial positive charge, resulting a formation of Polar Covalent Bond. Polar Molecules: A molecule is polar when their exists two separate poles of charges in the molecules at two different positions. Example of Polar Molecules: H2O, HCL, NH3,SO2 etc. Non –Polar Molecules: Those molecules in which there is no charge separation of positive and negative charges in a molecule and such a resultant molecule is neutral. Example of Non Polar Molecules: Co2, BF3, BeH2, CH4 etc.
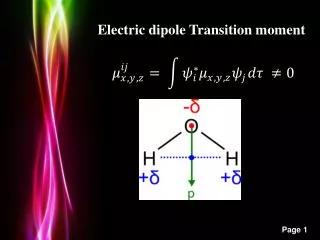
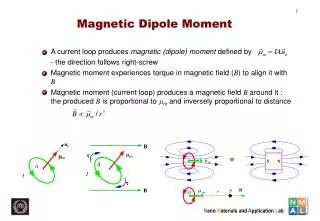
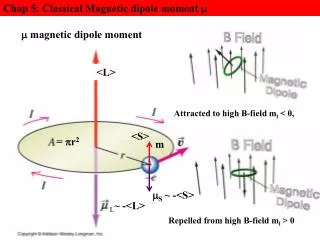
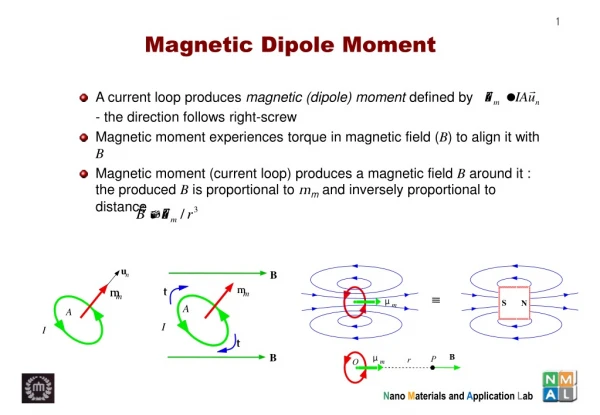
Dipole Moment • Definition: Dipole Moment refers to the quality of a system to behave like a dipole. It is defined as the product of the magnitude of charge on atoms and the distancebetween two bonded atoms. • Its common unit is Debye and SI unit is coulomb meter. • Formula: ц = Q * R, where Q is charge and R is distance between two bonded atoms. • It is a vector Quantity and its direction is given by an arrow pointing from positive to negative centers. • 1 Debye = 3.33564 * 10^-30 coulomb meters • Greater the charge larger will be the dipole moment • Smaller the distance lesser will be the dipole moment
Dipole Moment of Polar Covalent Bond When a bond is formed between two dis-similar atoms, then more electronegative element becomes more electron rich whereas the less electronegative element becomes electron deficient, resulting a polar Covalent Bond. The dipole moment of polar bond is non-zero and is represented as Цbond ≠ 0 The dipole moment of polar molecules is non- zero.
Dipole Moment of Non-PolarCovalent Bond When a bond id formed between two similar atoms with similar electronegativity, then bond pair of electrons is equally attracted by both the atoms. As e result both the atoms are neutral.Such a bond formed is Non-Polar Covalent Bond. The dipole moment id non- polar bond is zero and is represented as цbond =0. The resultant dipole moment of a non- polar molecule is zero and is represented as цmolecule =0.
Example of Dipole Moment H2O (Water): In Case of water, there are two bonds each one is polar. We can get the resultant of both O-H bonds is in upward direction by adding vector ally. Additionally, the one pairs increase the molecular dipole since they consists of significant negative ion construction. So we call water a polar molecule because it has a molecular dipole. BeCl2: In Case of BeCl2, two polar bonds at 180 degree from each other and of the same magnitude. Thus, they cancel each other. So molecule is non- polar even though the bond is polar. BF3: In BF3, three bond dipoles and each at 120 degree from each other. These add vectorially in such a way as to cancel each other out. So, the molecule is non- polar. CH4: In Methane molecule, there are 4- polar bonds oriented at the tetrahedral angle of 109.5 degree from each other and these form bond dipoles cancel out each other. So this molecule is non- polar. PCL5: In Phosphorus pentachloride, the 3 equatorial bond cancels out each other dipole moment and axial bonds are at 180 degree to each other and cancel Out. So, molecule is non- polar.