Alkanes: Structure and Conformation
Alkanes: Structure and Conformation. Compounds Contain Only C, H General Formula: C n H 2n+2 (Saturated) Common Source of Alkanes: Petroleum Separation Technique: Fractional Distillation Boiling Point (Size) Method of Separating Basic Building Block of More Complex Organics.

Alkanes: Structure and Conformation
E N D
Presentation Transcript
Alkanes: Structure and Conformation • Compounds Contain Only C, H • General Formula: CnH2n+2 (Saturated) • Common Source of Alkanes: Petroleum • Separation Technique: Fractional Distillation • Boiling Point (Size) Method of Separating • Basic Building Block of More Complex Organics
Alkane Shape: Straight Chains • “Straight Chain” Better Termed “Unbranched” • Actually Zig-Zag Structures (Tetrahedral Carbon Atoms) • All Carbons sp3 Hybridized
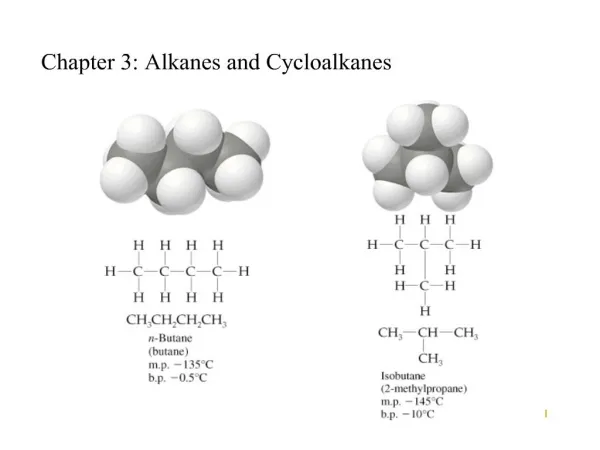
Branched Alkanes: Simple • Constitutional Isomers: Same Formula; Different Connectivity • Butane and Isobutane: C4H10 • Pentane, Isopentane, Neopentane: C5H12 • Different Properties: BP, MP, Density, Refractive Index etc. • Number of Constitutional Isomers Increases w/ # of Carbons
Alkane Nomenclature: The Rules Unbranched Alkanes
Alkyl Group Nomenclature Unbranched Alkyl Groups
Branched Alkanes • Locate Longest Continuous Chain (Parent Name) • Number Carbons in Chain; Begin @ End Nearest Substituent • Use Number of C on Parent Chain; Locate Substituents • Assign C Number to Each Substituent • Use Same C Number If Multiple Substituents • If Substituents Identical, Use Di-, Tri-, Tetra- Designations • Equal Length Chains Compete, Parent Chain Most Substituted • First Branches Equivalent; Choose Lowest Possible # Set
Branched Alkanes: Examples • Once Long Chain Found; Simplify w/ Alkyl Abbreviations
Branched Alkyl Groups • Can Name as Simple Alkane w/ “yl” Ending Replacing “ane”
Alkyl Halides • Alkyl Halides Named as “Halo”-Alkane • “Halo” = Fluoro, Chloro, Bromo, Iodo
Alcohols • Note the “-ol” Ending • Use Same Di-, Tri-, Tetra- to Indicate Multiple • We’ll Designate Alcohols as Priority Groups (Give Low #)
Monocyclic Alkanes/Alcohols Lowest Number Set • “Cyclo” Added Indicates Cyclic Structure
Terminal Alkenes Feature an “ene” Ending
Properties of Alkanes • Boiling Point Increases Regularly for Unbranched • Branching Lowers Boiling Points (Van der Waals, SA) • Melting Point in Unbranched Increases Regularly Within • ODD or EVEN Numbered Series (Not Both) • Density Less Than 1.0 g/mL (Less Than H2O) • Solubility: Quite Insoluble in H2O; Less Dense Float • Non Polar (Like Dissolves Like) • No Hydrogen Bonding
Sigma Bonds in Hydrocarbons • Alkane Sigma (s) Bonds Formed From sp3 Hybridized C • These Bonds Can Freely Rotate • Temporary Shapes Adapted via Rotation: CONFORMERS • Conformers can have Different Energies • Need to Have a System for Depicting Various Conformations • Newman Projections • Sawhorse Formulas
Newman Projections/Sawhorse Formula • R Groups 180° Apart (Anti Conformation) • 4 Atom Angle (3 Bond Angle) is a Dihedral (Torsional) Angle • Having Large Groups Anti is Low in Energy
Newman Projections/Sawhorse Formula • When R Groups 0° Apart (Ecclipsed Conformation) • Having Large Groups Anti is High in Energy • Can Adapt any Range of Conformations in Between
Conformational Analysis: Butane Energy • Can Plot these Points and Connect w/ Curve: PES
Cyclohexane Conformation • Cyclohexanes Adapt Chair Conformations (Boats and Others) • Ax: Axial (Straight up and Down on Chair) • Eq: Equatorial (Parallel to Next Bonds over in Chair)
Cyclohexane Conformation: Cis • Methyls are Cis (Same); Two Energy Equivalent Chairs • Ring Flips Interchange Chair Conformations
Cyclohexane Conformation: Trans • Methyls are Cis (Same); Two Energy Inequivalent Chairs • Diequatorial Much More Stable than Diaxial Conformation
Cyclohexane Conformation: Notes • With Multiple Same Substituents; More Equatorial = Better • Larger Groups Tend to Adopt Equatorial Positions • tert-butyl Groups Nearly Always Equatorial • Conformation Setters (Lock Ring) • Set Other Groups Relative to tert-butyl • Fused Rings (Decalin, for Example) Can be Drawn as Chairs
Reactions: Hydrogenation • Addition of Hydrogen (H2) Across a Multiple (p) Bond • Ethanol (CH3CH2OH) is a Common Solvent
Reactions: Alkyl Halide Reducation • Zn Transfers Electrons to C of Alkyl Halide • Alkyl Halide is Reduced (Reduction = Gains Electrons) • Zn: Good Two Electron Donor (Reductant, Reducing Agent)
Reactions: Alkylation of Terminal Alkynes • NaNH2 (-NH2) to Deprotonate Alkyne (Acid/Base Reaction) • Anion Reacts with Alkyl Halide (Bromide); Displaces Halide • Alkyl Group Added to Alkyne • Alkyl Halide Must be 1° or Me; No Branching at 2nd (b) Carbon