Efficacy of Antiangiogenic Thrombospondin-1 Peptides in Treating Canine Cancers
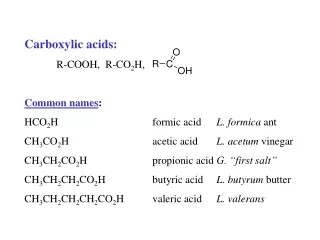
This study investigates the anti-cancer effects of Thrombospondin-1 peptides, specifically ABT-526 and ABT-510, in dogs with naturally occurring cancers, including soft tissue sarcomas. Conducted by a collaborative team from Animal Clinical Investigation, LLC and Abbott Laboratories, results from 242 cases show promising responses. Notably, 19 of 180 evaluable cases displayed objective responses, with a disease stabilization rate of 13%. Findings highlight the translational potential of using canine cancer models to inform human cancer therapies.

Efficacy of Antiangiogenic Thrombospondin-1 Peptides in Treating Canine Cancers
E N D
Presentation Transcript
ABT-526 and ABT-510, Antiangiogenic Thrombospondin-1 Peptides, Are Active in Dogs With Naturally Occurring Canine Cancers Including Soft Tissue Sarcomas Anthony Rusk1, Jack Henkin2, Evelyn McKeegan2, Fortuna Haviv2,Sandra Majest2, and Chand Khanna Animal Clinical Investigation, LLC1 and Abbott Laboratories2
Thrombospondin-1 CH CH CH CH 3 3 3 3 RGD H H H H S S O O O O H H H H C C C C CH CH CH CH H H H H C C C C OH OH OH OH CH CH CH CH 3 3 3 3 N N N N 3 3 3 3 3 3 3 3 EGF-like binding CH CH CH CH O O 3 3 3 3 O O O O O O O O O O O O I O O O O 3 3 3 3 O O H H H H H H H H _ H H H H H H H H - heparinbinding N N N N N N N N N N N N 1 1 NH2 1 N N N N 2 2 2 3 3 3 3 3 3 Procoll N N N N I I N N N N N N N N N N N N N N N N H H H H C C C C 3 3 3 3 H H H H H H H H H H H H O O O O O O O O O O O O O O O O CH CH CH CH S 3 3 3 3 H H H H C C C C cell 3 3 3 3 (trimer) H H H H C C C C binding H H H H C C C C 3 3 3 3 3 3 3 3 NH NH NH NH + + + + HN HN HN HN A-258526 A-258526 A-258526 A-258526 COOH NH NH NH NH 2 2 2 2 • Large multi-domain protein with multiple receptors • Anti-angiogenic domain is 2nd type-1 repeat • Drs. Noel Bouck, Olga Volpert, Jack Henkin • Series of related TSP-1 peptides: ABT-526
ABT-526 1 mg/kg/day ABT-526 10 mg/kg/day Vehicle ABT-526 Inhibits Mouse Corneal Neovascularization (b-FGF micropellet)
Non-Human Primate Beagle Dog Phase I Human Clinical Trials Small Animal Preclinical Tumor-Bearing Dog Studies Tumor-Bearing Dog Studies Integration of a Comparative Approach Phase II Human Clinical Trials Phase III Human Clinical Trials New Cancer Drug Activity Toxicity Pharmacokinetics Pharmacodynamics Dose Regimen Schedule Biomarkers Responding Histologies Combination therapies
TO PROVIDE OPPORTUNITIES TO INCLUDE NATURALLY OCCURRING CANCER MODELS IN THE STUDY OF CANCER BIOLOGY AND THERAPY Translational Opportunity • Cancer IN Companion Animals • 75 Million Companion Animals in the US • 6 million pet dogs diagnosed with cancer each year • Pet owners seek advanced care for their pets Companion Animal Cancer Models Large outbred Animals Strong Genetic similarities to Humans Naturally Occurring cancers Immune competant and syngeneic Relevant Tumor Histology/Genetics Relevant Response Profiles to Conventional Chemotherapy Tumor Heterogeneity Metastasis Biology Recurrence/Resistance
Predictions based on past Experience: Thalidomide Sedation at higher doses 12 cases treated No objective responses Disease stabilization 3 cases All cases measurable and advanced stage of disease Design:Open label single agent Eligibility: Histologically confirmed Measurable No concurrent cancer therapy Washout of 21 days Endpoints: Toxicity Pharmacokinetics (limited) Tumor Response Significant disease stabilization Objective response Thrombospondin-1 Peptides in Pet Dogs with Measurable Malignant Cancers Khanna et al ASCO 2002
TSP-1 Peptide Assignment, Dose, Schedule • Dog #1-26: ABT-526 12.5 mg BID, SC • Dog #27-72: ABT-526 0.5 mg/kg BID, SC • Dog# 1-30: ABT-510 12.5-17.5 mg BID, SC • Dog# 31-170: ABT-510 1.0 mg/kg QD or divided BID, SC
Animal Clinical Investigation, LLC: Cancer Network • Accrual Total: 242 Cases (2 years) Evaluable: 180 (68 sarcomas) *on therapy > 30 days • No Toxicity • Relevant population (Geriatric Dogs) • No arthritis • No delayed wound healing • No keratitis • Some dogs continue on therapy over 3 years Results: Accrual and Toxicity
Results: Pharmacokinetics / In Vitro Activity In Vitro Activity Serum Pharmacokinetics VEGF induced Endothelial Cell chemotaxis ABT-526 inhibited canine EC chemotaxis by greater than 60% at 100 nM concentrations. Detection limit 10 ng/ml 100nM = 0.1 µg/mL ABT-526 half-life = 48 min (range 33 to 60 min) ABT-510 half-life = 44 minutes (range 36 to 44 min) Pharmacokinetic profile stable after 1 month
RESULTS: RESPONSES • Significant stable disease seen in 23/180 (13%) evaluable cases • Objective responses: 19/180 (11%) evaluable cases • Head and Neck Carcinoma • Mammary Carcinoma • NH lymphoma • Cutaneous lymphoma • Sarcoma (Objective responses seen in 9/68 (13%) dogs • Synovial Sarcoma • Hemangiosarcoma • Soft Tissue Sarcoma
Day 0 Day 60 Day 30 Day 270 Day 90 Response: Maxillary squamous cell carcinoma (T3N0M0)
Day 0 Day 30 Day 60 Metastatic Synovial Sarcoma (Agent: TSP-1)
Predictors of Response?? • Response to therapy could not be predicted based on: • Initial tumor burden • Stage of disease • Primary versus metastatic lesions • Past treatments • Notable responses: • Slow Progression: Progression rate that would allow > 30 days of treatment • Soft tissue sarcoma (No responses in 10 dogs with osteosarcoma) • Lymphoma • Radiation “field’ failures
# * * * * Assessment of Biomarkers of Drug Exposure/Response Circulating endothelial cells (CEC’s) and CEC subsets may predict exposure and response to TSP-1
Thrombospondin-1 Peptides • Ask appropriate questions…Listen to the Answers • Antiangiogenic TSP-1 peptides are active against bulky and metastatic disease • Antiangiogenic TSP-1 peptides require extended time on therapy • Antiangiogenic TSP-1 peptides are active against NH-Lymphoma • Most dogs do not respond to therapy • Determinants of the responsive “patient” not known • Resistance has been seen in 80% of dogs responding to antiangiogenic therapy • The biology of antiangiogenic responses seen in measurable tumors may not predict responses against micrometastatic disease • Combination of TSP-1 peptides with cytotoxic chemotherapy are complimentary • Circulating endothelial cells may be a valuable predictor of response
Pet dogs Biomarker Validation -Circulating Endothelial Cells -Other Open Label Single Agent Any Measurable Malignant Cancer >500 dogs Adult Phase I Trial Adult Phase III Trial Adult Phase II Trial Adult Phase II Trial Pediatric Trials Soft tissue sarcoma (biomarker) Pet dogs Relapsed NH-Lymphoma Randomized Placebo Blinded Completed 105 cases Activity supported + CCNU Combination with Chemotherapy Biology Trials Pet dogs High Grade NH-Lymphoma Newly diagnosed Pet dogs Hemangiosarcoma Single agent adjuvant therapy Completed 30 case pilot No activity demonstrated TSP-1 + Radiation therapy -Head and Neck Carcinoma Translational Trials with Antiangiogenic Thrombospondin-1 Peptides
Ongoing Studies DOGS • Prospective study of antiangiogenic peptides of TSP-1 in dogs with measurable soft tissue sarcoma • Endpoints • Response (MRI confirmation) • Biomarkers of exposure and response (tumor, circulating cells, plasma) Goal: To define determinants of the response “patient” HUMAN • Over 400 cancer patients have been treated in Phase 1 and 2 trials with ABT-510 • Signals of efficacy is seen in soft tissue sarcoma • Characteristics of responding patient are unknow
Acknowledgements • Animal Clinical Investigation, LLC • Jennifer Turner • Kate Cadorette • Tony Rusk • Abbott Laboratories Inc • Jack Henkin • Evelyn McKeegan • Rick Lesniewski