Phases of Matter
250 likes | 745 Vues
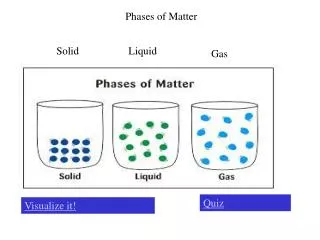
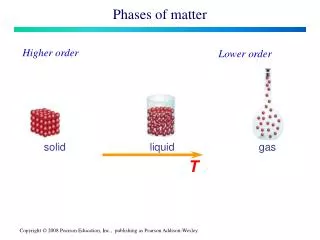
Phases of Matter. Chapter 10. Phases of Matter: are determined by the energy content and movement of the particles. What keeps the particles in liquids and solids together?. The movement of particles is directly related to the temperature of the substance,

Phases of Matter
E N D
Presentation Transcript
Phases of Matter Chapter 10
Phases of Matter: are determined by the energy content and movement of the particles
What keeps the particles in liquids and solids together? The movement of particles is directly related to the temperature of the substance, as the temperature cools the movement of the molecules slows and the molecules maybe attracted to each other. This is referred to as INTERMOLECULAR FORCES. Intermolecular forces are the attraction between molecules. These attractions are weaker than intramolecular forces like ionic and covalent bonds. The intermolecular forces can determine many of the physical properties of the substance. The stronger the intermolecular forces the higher the melting and boiling points of the substance. Why do you think this might be?
Why? The stronger the intermolecular forces the higher the energy required to disturb or break the attractions.
Types of intermolecular forces: Dispersion Forces (Van der Waals): Temporary attraction created as electrons orbit the nucleus. (-) (-) + + Positive nuclei will repel each other.
Dispersion Forces (Van der Waals): Temporary attraction created as electrons orbit the nucleus. (-) + (-) + When the electrons are concentrated on the same side as an exposed positive nuclei there will be an attraction. Creating a temporary bond between the two atoms. Types of intermolecular forces:
(-) + (-) + H H δ+ δ+ δ- δ- H H N N H H H H O O H H Types of intermolecular forces: Dispersion Forces (Van der Waals): Temporary attraction created as electrons orbit the nucleus. (weak attraction) Dipole – Dipole: The attraction between the oppositely charged ends of polar molecules. Hydrogen Bonding: The attraction created when a hydrogen atom is bonded to a highly Elecronegative atom (F, O, N). This attraction causes a higher than expected boiling point. (strong attraction)
Beginning State Ending State Process of Change How do substances change state? Solid Liquid Melting Solid Gas Subliming Liquid Solid Freezing Liquid Gas Evaporating/Boiling Gas Solid Deposition Gas Liquid Condensing
Location Altitude (ft) Pressure Boiling Point Boiling Points • The temperature at which a substance turns from liquid to vapor • Thought question: • Is the boiling point of a liquid the same in every location? NO! Boiling point is directly related to atmospheric pressure, and indirectly related to altitude! Sea Level 0 760 mmHg 100 ºC (373 K) Mt. Everest 29,028 240 mmHg 70 ºC (343 K)
Boiling vs. Evaporation Evaporation Occurs below the boiling point, only at the surface of the liquid. Occurs at the boiling point, throughout the entire liquid. Boiling Normal Boiling Point: the temperature at which vapor pressure of a liquid is equal to atmospheric pressure. Heat of vaporization: the amount of heat required to vaporize a liquid.
Freezing and Melting Points At what temperature does water freeze? SAME! 0 ºC (Freezing point) At what temperature does ice melt? 0 ºC (Melting point) Heat of Fusion: the amount of heat required to melt a solid to liquid
We often use graphs to show the correlation between vapor pressure and boiling. The graph below is the vapor pressure curve for four different substances. Using this graph we can find the “Normal boiling point” for any substance by observing its boiling point at standard pressure (101.3 kPa, 1 atm, 760 mmHg). For example the “normal boiling point” of substance A would be 35°C. What would the normal boiling of substance D be? 100°C Based on this information, what do you think substance D might be? Water
Line D represents water. If the atmospheric pressure in a flask is lowered to 70 kPa, water would boil at what temperature?
Line D represents water. If the atmospheric pressure in a flask is lowered to 70 kPa, water would boil at what temperature? 92ºC
Do the practice graph in your packet. • Answers: • 1. ~32kPa, ~7kPa (your answer may differ by +/- 1) • 2. 70ºC • 3. ~21kPa • 4. ~74ºC, ~115ºC
Particles are Always in Motion Kinetic-Molecular Theory GASES • Gases are composed of tiny particles • Particles are in constant motion • Elastic collisions occur between particles • There are no attractive forces between particles • Kinetic Energy increases when temperature increases. Ideal Gases!
Particles are Always in Motion Kinetic-Molecular Theory LIQUIDS • Viscosity: the friction or resistance to motion that exists between the molecules of a liquid • Surface Tension: the imbalance of forces at the surface of a liquid
Particles are Always in Motion Kinetic-Molecular Theory SOLIDS • Particles are “locked” in position and can only vibrate • Can be classified by three main characteristics: • Hardness • Electrical Conductivity • Melting Point
How do we classify solids? • Crystalline Solids: • organized, repetitive unit cells, sharp melting point • metallic: conduct electricity well • molecular: soft, low melting point • ionic: hard, brittle • covalent-network: conduct electricity at high temperatures • Amorphous Solids: • not “true” solids because molecules do move some • high viscosities make them appear solid • they soften before melting
For Next Class: • Complete homework page of packet.