Energy, Energy Transfer
590 likes | 1.38k Vues
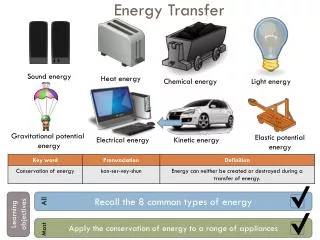
Energy, Energy Transfer. Working with Energy. Energy. Energy can be neither created nor destroyed , during a process it may change from one form to another but the same amount is present. Forms of Energy. Total Energy includes: Thermal Mechanical Kinetic Potential Electric

Energy, Energy Transfer
E N D
Presentation Transcript
Energy, Energy Transfer Working with Energy
Energy • Energy can be neither created nor destroyed, during a process it may change from one form to another but the same amount is present.
Forms of Energy • Total Energy includes: • Thermal • Mechanical • Kinetic • Potential • Electric • Magnetic • Chemical • Nuclear
Thermodynamics • Concept of Total Energy is hard to apply • Generally work with change in energy between two states
Energy • Macroscopic: forms of energy a system possesses as a whole based on an outside reference frame • Kinetic, potential • Microscopic: forms of energy related to the molecular structure of a system and degree of molecular activity and is independent of outside reference frames. • Sum of microscopic forms of energy is called internal energy
Energy • Internal Energy: symbol U • Kinetic Energy: symbol KE • KE = ½mV2 (kJ) (V is velocity) • ke = ½V2 (kJ/kg) • Potential Energy: symbol PE • PE = mgz (kJ) • pe = gz (kJ/kg) • Generally neglect magnetic, electric, surface tension energies
Energy • Total Energy: • E = U + KE + PE = U + ½mV2 + mgz (kJ) • e = u + ke + pe = u + ½V2 + gz (kJ/kg) • For a closed system with no change in elevation or velocity, called a stationary system, the change in energy is the change in internal energy U
Energy • For control volume: • Define the flow of mass per unit time as mass flow rate : Energy flow rate:
Internal Energy • Internal Energy: sum of kinetic and potential energies of molecules • Sensible Energy: translational, rotational, vibrational, spin kinetic energies • Latent Energy: energy related to phase changes • Chemical Energy: energy related to electron changes • Nuclear Energy: energy related to nucleus changes
Energy • Total energy is contained or stored in system, static forms of energy • Heat transfer and Work are energy not stored in system, dynamic forms of energy or energy interactions. Only recognized at the boundary of a system as they cross it • Note: do not confuse heat transfer and thermal energy
Mechanical Energy • Mechanical energy is the form of energy that can be converted to mechanical work completely and directly by an ideal mechanical device such as an ideal turbine.
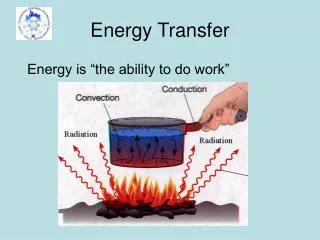
Heat is the form of energy transferred between two systems (or a system and its surroundings) by virtue of a temperature difference Energy Transferred by Heat
Heat • Heat is energy in transition, only identified as it crosses the boundary of a system
Heat • Symbol for heat is Q, q for per unit mass • Rate of heat flow per unit time is,
An adiabatic process is one that has no heat transfer occurring An adiabatic process is not necessarily an isothermal process Heat
Energy Transfer by Work • If the energy crossing the boundary of a closed system is not heat, it is work • Work is the energy transfer associated with a force acting through a distance • Work done between states 1 and 2 is, W12= F * d (kJ) • Work per unit mass is, w = W/m (kJ/kg)
Work done per unit time is called power, (kJ/s) or (kW) Energy Transfer by Work
Heat transferred to a system and work done by a system are positive Heat transferred from a system and work done to a system are negative Sign Convention
Heat and Work • Energy transfer mechanisms between a system and its surroundings: • Both are recognized at the boundaries of the system as they cross • Systems possess energy, but not heat and work • Both are associates with process, not state • Both are path functions
Differentials • Path functions have inexact differentials, δ • Heat and work are path functions • Differential amount of heat is δQ • Point functions have exact differentials, d • Properties are point functions • Differential amount of pressure change is dP
Electrical Work • Units, kilowatts, kW, kJ/s
Mechanical Work • W=Fs (kJ) • If force is not constant;
W = Fs =(T/r)(2πrn) = 2πnT (kJ) where T is torque applied r is radius n is number of revolutions (kW) Shaft Work
δWspring = F(x)dx F(x)=kx (kN) Wspring =½k(x22 –x12) (kJ) Spring Work
Work Done to Raise or Accelerate a Body • Calculate the change in potential or kinetic energy
First Law of Thermodynamics • Energy can be neither created nor destroyed during a process; it can only change forms • Also called conservation of energy principle
If process is adiabatic, the change in total energy of the system is equal to the net work First Law of Thermodynamics
If no work is done on or by the system, the change in total energy is equal to the net heat transfer First Law of Thermodynamics
The net change (increase or decrease) in the total energy of the system during a process is equal to the difference between total energy entering and the total energy leaving the system during the process Ein – Eout = ΔEsystem Energy Balance
Energy Change of a System • Energy change = Energy at final state – Energy at initial state • ΔEsystem = Efinal– Einital = E2 – E1 • ΔE = ΔU + ΔKE + ΔPE • ΔU=m(u2–u1) • ΔKE=½m(V22–V12) • ΔPE=mg(z2–z1)
Special Case • For a stationary system (no vertical movement or velocity) ΔKE = ΔPE = 0 so ΔE = ΔU
Mechanisms of Energy Transfer • Heat Transfer, Q • Work Transfer, W • Mass Flow, m • Ein–Eout= (Qin–Qout) + (Win–Wout) + (Emass,in–Emass,out) = ΔEsystem
Energy Conversion Efficiencies • Combustion Efficiency ηcombustion = Q/HV where: Q is the amount of heat released during combustion HV is the heating value of the fuel burned
Properties of Pure Substances Properties at a State
A substance with a fixed chemical composition throughout is called a pure substance Mixture of various chemical elements or compounds qualifies as a pure substance as long a the mixture is homogeneous Pure Substance
A mixture of two or more phases of a pure substance is still a pure substance as long as the chemical composition of all phases is the same. Pure Substance
Phases of a Pure Substance • Three principle phases, solid, liquid, gas • May have phases within principle phases, each with a different molecular structure • Carbon: graphite and diamond
Solid: rigid three-dimensional, fixed positions, strong bonds between molecules Liquid: not fixed, weaker bonds Gas: molecules far apart, weakest bonds Phases of a Pure Substance
Using water as an example Compressed liquid, sub-cooled liquid, a liquid that is not about to vaporize Add heat, at constant pressure, remains a compressed liquid until reaches 100°C Phase Change Process of Pure Substances
At 100°C becomes a saturated liquid, a liquid about to vaporize. Any heat added will cause some of the water to vaporize, but no change in temperature Phase Change Process of Pure Substances
As heat is added, becomes a saturated liquid-vapor mixture, temperature remains at 100°C. As more heat is added more saturated liquid becomes saturated vapor. If any heat is removed, saturated vapor converts back to saturated liquid Phase Change Process of Pure Substances
Heat is added until all the saturated liquid is converted to saturated vapor Phase Change Process of Pure Substances
As heat is added to the saturated vapor, it becomes superheated vapor, temperature increases above 100°C If heat is removes from superheated vapor, it reduces the energy level until it reaches saturated vapor level, then condenses Phase Change Process of Pure Substances
Saturation Temperature and Pressure • The temperature at which water starts boiling depends on the pressure; if the pressure is fixed, so is the boiling temperature. • For a given pressure, the temperature at which a pure substance changes phase is called the saturation temperature, Tsat • For a given temperature, the pressure at which a pure substance changes phase is called the saturation pressure, Psat
Phase Change Energy • Latent heat: the amount of energy absorbed or released during a phase change • Latent heat of fusion: amount of energy released during melting/absorbed during freezing • Latent heat of vaporization: amount of energy absorbed during vaporization/released during condensation