1 / 10
Télécharger la présentation 

Phase Changes
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
Phase Changes Courtesy www.lab-initio.com
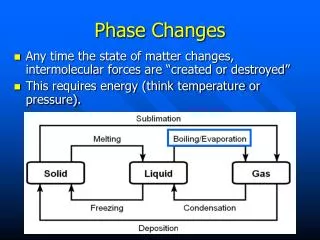
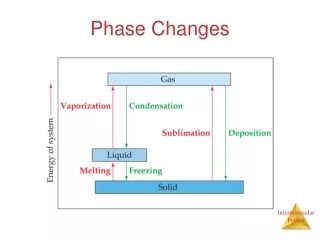
CA Standards • Students know energy is released when a material condenses or freezes and is absorbed when a material evaporates or melts.
constant Temperature remains __________ during a phase change. Water phase changes
Phase Diagram • Represents phases as a function of temperature and pressure. • Critical temperature: temperature above which the vapor can not be liquefied. • Critical pressure: pressure required to liquefy AT the critical temperature. • Critical point: critical temperature and pressure (for water, Tc = 374°C and 218 atm).
Phase Diagram for Carbon dioxide Carbon dioxide
Carbon Phase Diagram for Carbon
More Related