Heat Changes
Heat Changes. Enthalpy. E N T H A L P Y - H. heat content in matter:. E N T H A L P Y - H. heat content in matter: natural systems tend to go from a state of high energy to a state of low energy. E N T H A L P Y. The phases of matter represent "classes" of molecular motion found

Heat Changes
E N D
Presentation Transcript
Heat Changes
E N T H A L P Y - H heat content in matter:
E N T H A L P Y - H heat content in matter: natural systems tend to go from a state of high energy to a state of low energy.
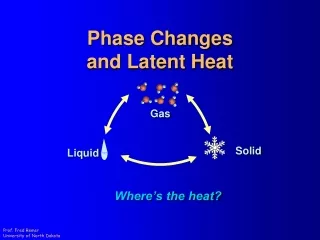
E N T H A L P Y The phases of matter represent "classes" of molecular motion found at different temperatures. Solid Gas Liquid
E N T H A L P Y Kinetic Energy X The phases of matter represent "classes" of molecular motion found at different temperatures.
E N T H A L P Y HEAT X The phases of matter represent "classes" of molecular motion found at different temperatures.
E N T H A L P Y q = U + P V q = heat gained or lost U = internal energy P = Pressure V = volume
E N T H A L P Y Hfus - heat of fusion As energy is added to a solid at its melting point, ALL the energy is used to increase the kinetic energy of the molecules during the phase change.
E N T H A L P Y Hfus - heat of fusion The amount of heat needed to change one gram of a solid into a liquid at its melting point.
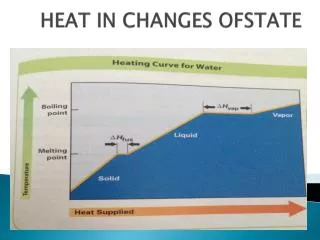
Temperature Rise Heat Added
Every phase change is accompanied by a change in the energy of the system.
As the temperature of a solid increases, its particle vibration increases.
When a solid melts, its particles are freed to move with respect to one another.
This melting process is called fusion.
The increased freedom of motion of the molecules or ions comes at a price...
measured by the heat of fusion, or enthalpy of fusion, DHfus
As the temperature of a liquid increases, the molecules move with increasing energy.
When increasing temperature, the concentration of gas-phase molecules just above the surface of the liquid increases.
These gas-phase molecules exert a pressure on the liquid called vapor pressure.
This vapor pressure increases with temperature until it equals the atmospheric pressure.
This is the boiling point of the liquid.
The energy required to cause this transition..
is called the heat of vaporization or enthalpy of vaporization, DHvap
solid/liquid phase change q = m Hfus q = heat gained or lost m = mass DHfus = heat of fusion
E N T H A L P Y Hfus H2O = 334 J/g Hfus lead = 24.72 J/g
E N T H A L P Y As energy is added to a liquid at its boiling point, ALL the energy is used to increase the kinetic energy of the molecules during the phase change.
E N T H A L P Y Hvap- heat of vaporization The amount of heat needed to change one gram of a liquid into a gas at its boiling point.
liquid/gas phase change q = m Hvap q = heat gained or lost m = mass DHvap = heat of vaporization
E N T H A L P Y HvapH2O = 2260 J/g
change temp within phase q = m TCp q = heat gained or lost m = mass DT = “change in temperature” Cp = specific heat capacity
Specific Heat Capacity The heat required to raise the temperature of one gram of a substance by one Celsius degree.
Specific Heat Capacity Cp of ice = 2.06 J/g.Co Cp of water = 4.18 J/g.Co Cp of steam = 2.02 J/g.Co Cp of copper = .385 J/g.Co
q=mTCp - change temp q=mHvap - change phase q=mTCp - change temp q=mHFUS- change phase q=mTCp - change temp
Sixteenth Lab Heat of Fusion of Ice
Change temp of ice Melt ice Change temp of liquid water Vaporize water Change temp of vapor Add total heat used How much heat is needed to convert 250 grams of ice at -30 oC to vapor at 150 oC?
Step 1 raise temp of ice q = m DT Cp q = (250g) (30 Co) (2.06 J/g. Co) q = 15,450 Joules
Step 2 melt ice q = m DHfus q = (250g) (334 J/g) q = 83,500 Joules
Step 3 raise temp of liquid water q = m DT Cp q = (250g) (100 Co) (4.18 J/g. Co) q = 104,500 Joules
Step 4 vaporize liquid water q = m DHvap q = (250g) (2260 J/g) q = 565,000 Joules
Step 5 raise temp of water vapor q = m DT Cp q = (250g) (50 Co) (2.02 J/g. Co) q = 25,250 Joules
Step 6 q = 15,450 83,500 104,500 565,000 25,250 q = 794,000 Joules
What is Specific Heat Capacity
Which has the greater? Specific Heat Capacity H2O Fe or
A Calorimeter is used to measure heat change
Seventeenth Lab Specific Heat of a Metal
Eighteenth Lab Lab Burners and BTUs
Enthalpy end