Hydrogen electrolysis
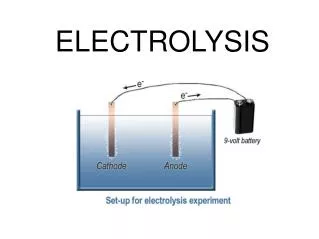
Hydrogen electrolysis. Hydrogen electrolysis is the process of running an electrical current through water (H 2 O) and separating the hydrogen from the oxygen. Process is the REVERSE of what occurs in a fuel cell. (-) (+). 9v. + / -. e. -. e. -. e. -. H 2. O 2. e. -.

Hydrogen electrolysis
E N D
Presentation Transcript
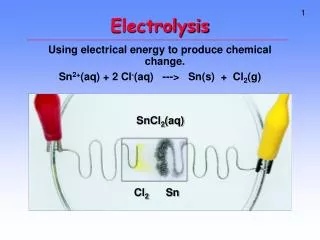
Hydrogen electrolysis Hydrogen electrolysis is the process of running an electrical current through water (H2O) and separating the hydrogen from the oxygen. Process is the REVERSE of what occurs in a fuel cell (-) (+)
9v + / - e - e - e - H2 O2 e - Electrolysis Cell Fuel Cell H2 O2 (H2O) H2O Anode Cathode Anode Cathode Fuel cell: Hydrogen and oxygen gases flow into the cell producing water and electricity. Electrolysis cell: Hydrogen and oxygen gases produced when electricity flows through an electrolyte containing water.
Electrochemical reactions • Cathode – hydrogen reduced • 4H2O + 4e- 2H2 + 4OH- • Anode – hydrogen oxidized • 2H2O O2 + 4H+ + 4e- • Overall reactions • 2H2O O2 + 2H2 • 4OH- + 4H+ 4H2O • Overall reaction requires electrical energy added
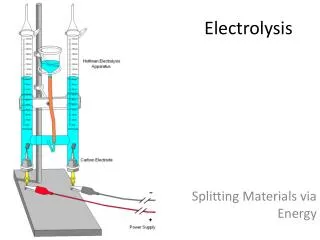
Today’s Lab Investigate bench top scale hydrolysis to produce Hydrogen Fuel


















![[READ DOWNLOAD] Hydrogen Production: by Electrolysis](https://cdn7.slideserve.com/12657103/read-download-hydrogen-production-by-electrolysis-dt.jpg)