CHEMICAL BONDING
CHEMICAL BONDING . Why bond? Atoms try to acquire particularly stable electron configuration (i.e the configuration maximizing binding energy) for their outer electrons form ions or molecules.

CHEMICAL BONDING
E N D
Presentation Transcript
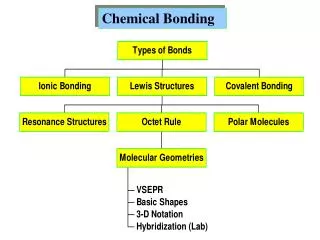
CHEMICAL BONDING • Why bond? • Atoms try to acquire particularly stable electron configuration (i.e the configuration maximizing binding energy) for their outer electrons form ions or molecules. • Most stable configuration for outer electrons is noble gas configuration (octet structure) ns2np6 • strategies to achieve more stable configuration: • give away electrons • accept electrons • share electrons • chemical bond is formed when it is energetically favorable, i.e., when the energy of the bonded atoms is less than the energies of the separated atoms. • Quantities which play a role in chemical bonding: • ionization energy (the energy required to remove an electron from a neutral atom) • electron affinity (the energy change when a neutral atom attracts an electron to become a negative ion) • electronegativity (the ability of an atom in a molecule to draw bonding electrons to itself) • types of bonds: • ionic bond • covalent bond • metallic bond
IONIC BOND • atoms achieve noble gas configuration by giving up or accepting electrons (usually electron transfer from metal to non-metal) formation of ion; • chemical bond is formed due to electrostatic attraction between two oppositely charged ions; • bond is strong, but becomes quickly weak when ion is displaced; materials usually brittle (e.g. glass, rock, egg shells,..) • compounds formed by ionic bond: e.g. NaCl, CaCl2,.. • examples of ions with noble gas configuration:
METALLIC BOND • metals have low ionization energy atoms give up outer (valence) electron, to be shared by all • metallic lattice = positive ions at fixed positions, in “sea” or “gas” of mobile electrons; • electron gas has “pressure” due to Pauli principle • electrons mobile good thermal and electrical conductivity • positive ions not free to move, but can vibrate • electron not tied down in particular bond can absorb and re-emit light over wide frequency range good reflector • bond is “elastic” since attraction due to mobile electrons bond holds even if ions displaced metals are malleable • ability of the electrons to spread between the cations and neutralize their charges metal ions ions can pack closely together; closeness of the packing of the atoms high densities of metals. • In some sense, piece of metal is like an extremely large molecule
Covalent Bond • COVALENT BOND • well-defined cluster of neighboring atoms share electrons - form “molecule” • state with shared electrons has lower energy than individual atoms • “valence electrons” = outer electrons • group number = number of valence electrons • “valence of element” = number of electron pairs shared to complete octet of electrons • examples: H2 , O2 , N2 • in H2, H atoms share one electron pair; • n O2, O atoms share two electron pairs; • in N2, N atoms share three electron pairs; • single H, O, N (“in statu nascendi”) are much more reactive than pair • Carbon • Carbon's outer electron configuration is4s2 4p2 it needs 4 electrons to complete octet shares 4 pairs of electrons • e.g. methane (“swamp gas”) • possibility of 4 covalent bonds large variety of possible compounds with C organic chemistry
Polar bonds • POLAR BONDS • Electron pairs shared between two different atoms not necessarily shared equally - sharing ratio depends on electronegativity; • “electronegativity” = ability of atom to attract an electron to itself; • electronegativity increases from left to right (i.e. grows with group number) in every row of the periodic table; • alkali metals (group 1) are least electronegative, halogens (group 7) are most electronegative; • non-polar bond = bond in which electrons are shared equally • polar covalent bond = bond in which one of the atoms exerts greater attraction for the electrons than the other (has larger electronegativity) • if difference in electronegativity is large enough, bond becomes ionic bond • GEOMETRY OF BONDS • electron-pair repulsion rule (EPRR): • Electron pairs surrounding an atom (be they shared or un-shared with other atom) repel each other and are directed to be as far apart as possible. (example: water molecule)
INTER-MOLECULAR FORCES • electric dipole forces: polar molecules exert electric dipole forces on each other; • e.g. water molecule: H(+) attracted to (-) partner of other molecule (oxygen of other water molecule, or solute constituent) “hydrogen bond”; • hydrogen bond is reason for water to be liquid at “normal” temperatures (note e.g. CO2 , CH4 are gases!) • Van der Waals forces: • fluctuations, shifts of charges within covalent molecule temporary dipole moment electrostatic dipole forces; • charge unbalances small forces weak (Van der Waals binding energies are 10-2eV ) • most liquids held together by VdW forces, • cohesion, surface tension
Water • water molecule: • oxygen in water has 4 electron pairs, 2 shared and 2 unshared; • EPRR e-pairs point to corners of tetrahedron, with O atom in center of tetrahedron, • H atoms sit at two of the corners of the tetrahedron; constituents of water form triangle; angle between the two lines from O to H is 105o (109o predicted from this model); • “Mickey Mouse shape” of water molecule • O is more electronegative than H water is polar molecule, more negative near O atom, more positive near H atoms. • polarity of water molecule good solvent
Why ice is less dense than liquid water • water is most dense as a liquid and least dense as a gas; solid water (ice) is less dense than liquid water, but more dense than a gas; • water vapor (gaseous water) is made up of individual water molecules that are not hydrogen bonded to other water molecules; • cooling, pressure hydrogen bonds form liquid; but in liquid, hydrogen bonds keep forming and breaking up; on average every water molecule in 3.4 H-bonds • at 4oC, water molecules as tightly packed as they will go • below 4oC , more and more stable hydrogen bonds form, eventually reaching 4 per molecule; the hydrogen bonds push the water molecules further apart, making it less dense ice floats on water