3.11 Phenols
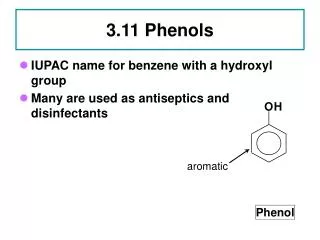
O. H. aromatic. Phenol. 3.11 Phenols. IUPAC name for benzene with a hydroxyl group Many are used as antiseptics and disinfectants . 3.12 Nomenclature for Phenols. Phenol 2-methylphenol, not 3-hydroxytoluene 3-chlorophenol. O. H. Phenol. 3.13 Physical and Chemical Properties.

3.11 Phenols
E N D
Presentation Transcript
O H aromatic Phenol 3.11 Phenols • IUPAC name for benzene with a hydroxyl group • Many are used as antiseptics and disinfectants
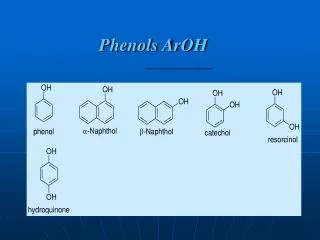
3.12 Nomenclature for Phenols • Phenol • 2-methylphenol, not 3-hydroxytoluene • 3-chlorophenol O H Phenol
3.13 Physical and Chemical Properties Water soluble High m.p. & b.p. Acid properties – acidic (~pH=4) Do not dehydrate Are not easily oxidized Do not substitute Cl with Lucas reagent
Hydrogen bonding in phenol m.p.= 40-43oC b.p. = 182oC In comparison, toluene m.p.= - 95oC b.p. = 111oC
3.13 Physical and Chemical Properties Water soluble High m.p. & b.p. Acid properties – acidic (~pH=4) Do not dehydrate Are not easily oxidized Do not substitute Cl with Lucas reagent
Acid properties of phenol pH = 4 pKa = 10
3.13 Physical and Chemical Properties Water soluble High m.p & b.p. Acid properties – acidic (~pH=4) Do not dehydrate Are not easily oxidized Do not substitute Cl with Lucas reagent
3.14 Uses • Many are used as antiseptics and disinfectants • - 2-phenylphenol is in Listerine
3.14 Uses (cont.) • 2. Polyphenolics are antioxidants- flavinoids • epicatechin found in green tea