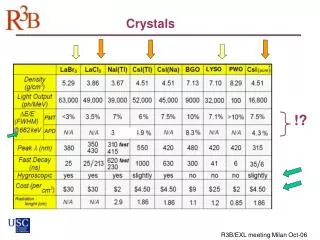
Crystals
Augusta High School. Crystals. Hannah Cole Jamison Irvine. 9th Grade February 26th, 2002. Table of Contents. Section Slide Number Introduction 3 Background Information on Topic 4 Experiment 6 Data and Graphs 16 Discussion and Data Analysis 17 Conclusion 19

Crystals
E N D
Presentation Transcript
Augusta High School Crystals Hannah Cole Jamison Irvine 9th Grade February 26th, 2002
Table of Contents Section Slide Number Introduction 3 Background Information on Topic 4 Experiment 6 Data and Graphs 16 Discussion and Data Analysis 17 Conclusion 19 Acknowledgments 20 References 21
Introduction • Problem: What type of Liquid, water or vinegar, will form the best type of crystal with what type of solid, salt or sugar. • Hypothesis: Water is a better liquid than vinegar. And salt is a better solid than sugar. • Research: Certain amount of water and certain amount of vinegar needed. Some will have a certain amount of sugar or salt added.
Background Information on Topic • Formula for Sugar is C6 H12 O6. • Formula for Salt is NaCl. • Formula for Vinegar is C4 H8 O2. • Salt is and Ionic Crystal. • Sugar is made up of atoms, which form together to make molecules. • The Ration between liquid and solid should be =. • Heating the liquid is needed to make the experiment work correctly, otherwise the solid will not be absorbed.
More Background Information • Snow and ice crystal are formed when a gas or liquid gets cold enough. • Quartz and other minerals from when the get hot enough. • Volcanic crystals form when the volcanic rock cools. • Crystals grow from solutions, when the substance is dissolved in a solution and start connecting together in the orderly arrangement of a crystal. • Seven Crystal Systems, Triclinic, Monoclinic, Orthorhomic, Tetragonal, Rhombohedral, Hexagonal, and Cubic.
Expirement • Materials • 2 Medium Sized saucepans • 5 16 ounce Glasses • 1 Measuring Glass • 1 cup Vinegar • 1 2/3 cup Water • 2/3 cup Sugar • 1 2/3 cup Salt • 5 Pencils • 5 4 inch pieces of string • 5 Stirring Spoons • 5 Paper Clips
Expirement • Procedure • Tie paperclip to string and then tie to pencil • Place water and vinegar in saucepans, Bring to boil • While liquids boil, label glasses. • Measure 1/3 cup boiling water and pour into glass, set aside. • Measure 1 cup boiling water, add 1 cup salt stir, set aside. • Measure 1 cup boiling water, add 1/3 cup water, stir, set aside • Measure 2/3 cup boiling vinegar, add 2/3 cup salt, stir, and set aside. • Measure 1/3 cup boiling vinegar, add 1/3 cup sugar, stir, set aside. • Put pencil, with string attached, on the rim of each glass. • Clean up, put glasses where they can be observed but not bothered.
Expirement • Control: Water • Dependent: Size and amount of crystals produces. • Independent: Types of crystals, vinegar, water, salt, sugar. • Constant: The procedure in which the experiment was performed and the time each was given. • Data: We collected data based on the appearance of each glass or type of crystal. We will list the changes that are made in the growth from day to day. • Trials: We only did one trial because of the amount of time that crystals take to grow.
Discussion And Data Analysis • Expected water/salt to grow best • Water/salt formed first and fastest • Vinegar/salt turned the paperclip and the crystal black • Vinegar/sugar grew up the string a little • Data supported hypothesis • Water/salt formed a film on the inside of the glass, and gathered at the top of the glass. • No graph, insufficient information/data. Data Table based on appearance.
Discussion And Data Analysis • Things that would have been different… • More trials • Used epsom salt instead of table salt • Use the same amount of all items/materials • Be more exact. • Find a new way of measuring the size of crystal, rather than just appearance.
Conclusion • What liquid (water or salt) forms the best crystal our of what solid (salt or sugar)? • Water was best for a salt crystal • Vinegar was best for a sugar crystal • Water/salt formed best mixture for, it formed the biggest and best crystal the fastest. • Vinegar/salt • Vinegar/sugar • Water/sugar • Water (control)
Acknowledgments We would like to thank our parents for supplying us with the things that we needed. We would like to thank Mr. Radloff for helping us with anything that we needed help with or didn’t understand. We also would like to thank any other people that helped us along the way. Thanks
References World Book, 17, Salt, Chicago, World Book Inc., 2002, 72-75 World Book, 18, Sugar, Chicago, World Book Inc., 2002, 959- 961 The New Book of Knowledge, 3, Chicago, Groiler incorporated, 1991, 591-593 The Merick Index, twelfth edition, White House Station, NJ., Merick and Co. Inc., 1996, page 641 and 1474 www.webmineral.com/crystal.shtml www.webmineral.com/chmical.shtml www.webmineral.com/specimens.shtml www.google.com/search?hl=en&crystals+of+sugar www.google.com/search?hl=en&crystals+of+vinegar