GEL ELECTROPHORESIS:
GEL ELECTROPHORESIS:. The method for separation and purification of nucleic acids and proteins. Electrophoresis of Onion DNA. The purpose of this lab is to first isolate onion DNA by lysis and then separate and isolate the individual nucleic acids of the onion DNA by gel electrophoresis. .

GEL ELECTROPHORESIS:
E N D
Presentation Transcript
GEL ELECTROPHORESIS: The method for separation and purification of nucleic acids and proteins.
Electrophoresis of Onion DNA • The purpose of this lab is to first isolate onion DNA by lysis and then separate and isolate the individual nucleic acids of the onion DNA by gel electrophoresis.
PART ONE: What is DNA?
BASES • DNA is composed of what are called bases known as purines and pyrimidines. • Each of these purines and pyrimidines are heterocyclic amines, therefore are basic in nature.
Purines ADENINE GUANINE Pyrimidines CYTOSINE THYMINE BASES OF DNA
HOW DO THESE BASES ‘HOOK’ TOGETHER? • It takes the additional bonding of a sugar and phosphate group.
THE SUGAR OF DNA 2-DEOXY-D-RIBOSE
SUGAR + BASE • The monosaccharide sugar 2-deoxy-D-ribose bonds with the purine base through carbon number 1 on the sugar and nitrogen number 9 on the base. • This linkage of a sugar and a base is known as a nucleoside.
ADDITION OF THE PHOSPHATE GROUP When the phosphoric acid forms a bond with the CH2OH it forms a phosphate ester with the nucleoside. The result is what is known as a nucleotide.
BASE + SUGAR + PHOSPHORIC ACID NUCLEOTIDE
The Big Picture • These nucleotides bond together to form a chain. • This chain is called a nucleic acid which is the backbone of DNA. • The bases of one nucleic acid chain bonds with the bases of another nucleic acid chain.
In the bonding of bases in a nucleic acid, a pyrimidine must be opposite a purine. For example: Thymine + Adenine Cytosine + Guanine The formation of these base pairs is done by hydrogen bonding, which in turn hooks two nucleic acid chains together. Although……
Part 2: What is Gel Electrophoresis?
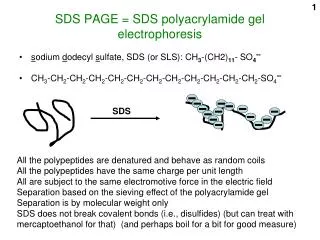
GEL ELECTROPHORESIS • Gel electrophoresis is used to separate proteins and in the case of this lab, nucleic acids, by relying on the movement of charged particles in an electric field. • The medium used is agorose gel submerged in a buffer solution. • A positive electrode is placed on one side of the buffer solution and a negative on the other.
The buffer solution should be either more acidic or more basic than the isoelectric point of the nucleic acid in question. This determines toward which electrode the nucleic acid will migrate in the arogose gel. If the nucleic acid’s PH corresponds to that of the buffer, there will be no net movement. more acidic negative electrode more basic positive electrode Equal no net movement Gel Electrophoresis Cont…..
MOVEMENT • Because a nucleic acid incorporates different side chains two different nucleic acid chains will have slightly different net charges at a particular PH. • Thus, their movement in the agorose gel will be different and electrophoresis can be used to separate different nucleic acids.
Nucleotides, at a PH of 7.4, ionizes the phosphate links between each nucleotide.This gives the DNA fragments a negative charge and causes them to migrate to the positively charged electrode.
Size is also a determining factor of how far a nucleic acid will migrate in the agorose gel. Larger polynucleotides move more slowly through the gel than smaller ones.
SODIUM DODECYL SULFATE (SDS) • SDS is used in gel electrophoresis because it binds to the nucleic acid causing it to unfold into a ‘rod like’ shape. Therefore, all of the nucleic acids because they have similar shapes, will tend to travel at rates proportional to their chain lengths. • Also, the SDS molecule ensures that the nucleic acids are negatively charge and that they will migrate toward the positive electrode.