Understanding Polyatomic Ions: Definition, Examples, and Charge Balancing
This text provides an insightful overview of polyatomic ions, contrasting them with monatomic ions. It starts by defining monatomic ions, explaining how they carry positive (cations) or negative (anions) charges based on the balance of electrons and protons. The focus then shifts to polyatomic ions, which are groups of two or more covalently bonded atoms that carry a charge. An example of a polyatomic ion, the nitrate ion (NO3–), is discussed in detail, including how to balance charges in compounds like magnesium nitrate (Mg(NO3)2).

Understanding Polyatomic Ions: Definition, Examples, and Charge Balancing
E N D
Presentation Transcript
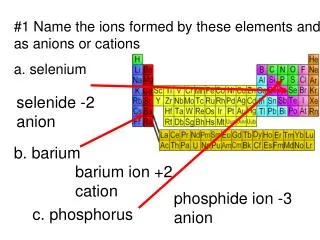
What are POLYATOMIC IONS? Lets first review what a monatomic ion is What is a group of atoms with charge? back to naming compounds
More electrons than protons = negative charge (anions) + + + + + Less electrons than protons = positive charge (cations) + + + + + In a single atom (monatomic), charge results from having either more electrons or less electrons than protons.
Mg 2+ Example: The magnesium ion has two less electrons than protons
Mg 2+ back to polyatomic ions Example: The magnesium ion has two less electrons than protons
O N O O
O N O O In a polyatomic ion, two or more atoms are covalently bonded together.
1– O N O O Together, they carry a charge
1– O N O O The nitrate ion (NO3–) has, in total, one more electron than protons (count them! 31 p+, 32 e-)
1- 1- 2+ O O N N O O Mg O O When balancing charges in an ionic compound, the polyatomic ion acts as a single unit of charge: Mg(NO3)2
O O N N O O Mg O O 1- 1- 2+ Mg(NO3)2 2+
O O N N O O Mg O O the charges are balanced 1- 1- 2+ Mg(NO3)2 2+ 2-
back to first slide back to naming compounds previous slide