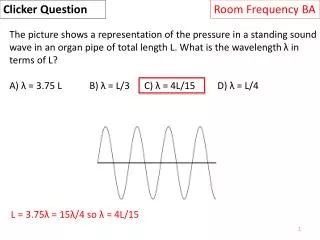
Clicker Question
Room Frequency BA. Clicker Question. When people breathe helium and then speak, their voices sound much higher in pitch! Given that a person’s throat and vocal chords, which produce the sounds, do not change, how does the speed of sound in helium compare to that in regular air?


Clicker Question
E N D
Presentation Transcript
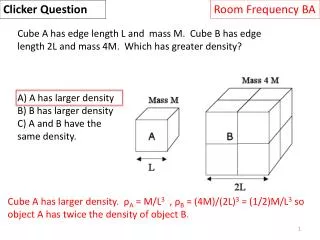
Room Frequency BA Clicker Question When people breathe helium and then speak, their voices sound much higher in pitch! Given that a person’s throat and vocal chords, which produce the sounds, do not change, how does the speed of sound in helium compare to that in regular air? sound in helium is faster sound in helium is slower sound has the same speed in air and helium; the change in pitch is from a different effect. v = fλ: since λ is determined by the throat size, and pitch goes up, f is also larger so v is larger (by about a factor of 3!)
Announcements • CAPA assignment #15 is due on Friday Dec 9th at 10 pm. • This week: Review in Section, Make-up Labs arranged in other sections. Last chance to make up labs! 5 labs must be completed in order to pass this course.
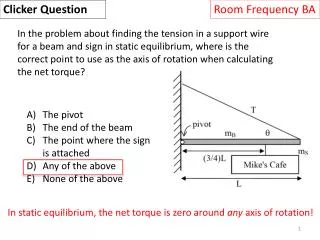
Final Exam News • Final Exam is Tuesday morning, Dec 13, 10:30am – 1pm • Exam will be held in Coors Event Center, on the West (mountain) side of the arena. Calc-based physics is on the east side; don’t sit down in the wrong side! • We are in sections 20, 21, 22, with individual seat assignments. Your seat assignmentwill be posted on CU Learn. • Bring student ID. We will be checking that people are in their assigned seat. • Practice Exams and Formula sheet are posted on CULearn; you can ignore Prob. 37 on the practice exam • Prof. Nagle will hold a review session on Thursday evening, Dec 8th, 7-9pm, in this room (Duane G1B30).
Temperature and Thermal Energy • Thermal Energy is in the KE and PE of the individual atoms in matter from their individual random motion! • Temperature is a measure of the thermal energy per atom, not the total thermal energy of a piece of matter • Conversion formulas: • We measure temperature by letting thermal energy “flow” between the thermometer and the system whose temperature we are trying to measure. • When the flow stops, we say the thermometer and system have reached thermal equilibrium. At this point the average thermal energy of the thermometer atoms is equal to the average thermal energy of system atoms. • This or any transfer of thermal energy is called heat.
How is Heat Transferred? • Heat is transferred mainly through 3 different mechanisms: • Conduction: Heat transfer direct inter-atom interaction without bulk movement of atoms. • Convection: Heat transfer by bulk movement of atoms, either forced (for example, stirring) or natural (boiling, magma) • Radiation: Heat transfer by electromagnetic radiation (light, especially infrared part of the spectrum)
Units of Heat • Heat is measured by a change in temperature, so the standard unit took a common material (water) and related the heat to the change in temperature of a certain amount of water: • 1 calorie (cal) is the amount of heat required to heat 1 gram of water 1 degree Celsius. • 1 kcal heats 1 kg of water by 1 °C • Warning: US “food energy” calories are actually kcal ( = 1 Cal) • British Thermal Unit (Btu) is 1 lb of water up 1 °F • The calorie is not the SI unit of energy! Using transfer of energy by stirring (mechanical action), one measures that 1 cal = 4.186 J
Heat: Thermal Energy Transfer • Heat is the thermal equivalent of mechanical work. It is a transfer of energy which changes the energy of the system. • The amount of heat transferred to some mass m is proportional to its temperature change ΔT. • For the same heat, different materials will have different changes in temperature ΔT: the structure of the matter determines how much thermal energy an atom can absorb • We define a quantity called the specific heat or heat capacity cwhich quantifies how much heat corresponds to a change in temperature of a given amount ΔT for unit mass. • Specific heats depend on material and the phase of the material: gas, liquid, solid, and even solid structure (crystal) • Heat Q = mcΔT
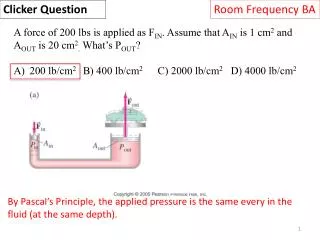
Room Frequency BA Clicker Question A kg of aluminum and a kg of iron at the same initial temperature receive the same heat Q. The final temperature of the iron is higher than that of the aluminum. What is the relation of their specific heats, ciron and caluminum? ciron>caluminum ciron = caluminum ciron < caluminum Q = mcΔT: since Q was the same for each and ΔT was larger for iron, its heat capacity must be smaller
Specific Heats • Generally specific heats of gases and liquids are much greater than those of solids • Specific heats can also depend on temperature, not just phase • Watch out for units! cal/(g°C) = kcal/(kg °C) = 4.186 kJ/(kg °C) • Liquid water has one of the largest heat capacities! • Humans (= water): Their temperature doesn’t change fast • Scalding fluids burn badly; they hold lots of thermal energy! • Combining materials at different temperatures and finding final temperature is called calorimetry. Used to measure heat released in chemical reactions.
Heating Power Example We want to change the temperature of the air in a room of volume 5 mx 4 mx 5 m with a heater which can deliver 1500 W. The initial temperature of the air is 15°C and we want the final temperature to be 20°C. How long will it take? First: What is the heat Q for this ΔT? Using ρair =1.29 kg/m3 and cair = 1050 J/(kg°C) we get Q = 6.8 x 105 J Q = (Heating Power)*(Δ time) so Δtime = Q/(Heating Power) Time to heat is 6.8 x 105 J/1500 W = 450 s = 7.5 min
Calorimetry Problems • A calorimeter is an insulated, sealed chamber that does not let heat or matter flow in or out • Often in simple problems, they do not mention using a calorimeter => so you must assume matter or heat flow out of system can be neglected!!! • Thermal energy by simple conduction always flows from hot to cold! This is independent of the conservation of energy… • Problem solution is based on energy conservation: heat lost by hot object must equal heat gained by cold object
CalorimetryExample 1 kg of gold at temperature 100°C is placed in a container of 1 liter of water at 25°C. What is the final temperature of the gold and water? Qgold=mgoldcgold(Tf-100) cgold = 130 J/(kg°C) Qwater=mwatercwater(Tf-25) cwater = 4186 J/(kg°C) Using ρwater =1000 kg/m3 and 1 liter = 1000 cm3 = 0.001 m3 we have mwater = 1kg |Qgold| =mgoldcgold(100 – Tf) = |Qwater| =mwatercwater(Tf-25) Solve for Tf ! You’ll get Tf = 27.26°C Is the final T between 25 and 100? Yes!
Latent Heats • Sometimes heat transfer does not change the temperature! • Usually the energy is causing a change in phase of the material, for example solid to liquid, or liquid to gas, but sometimes internal solid structure is changing. • The phase changes occur at some particular temperature, for example water changes from solid to liquid at 0°C and from liquid to gas at 100°C • Since the temperature doesn’t change, these are called latent heats and correspond to the amount of heat to change a certain amount of material of mass m between one phase and the other: Q = mL • Units of L are just kcal/kg ( = 4186 J/kg) • In calorimetry problems, you must account for energy to change phase of something if you reach its temperature of a phase change
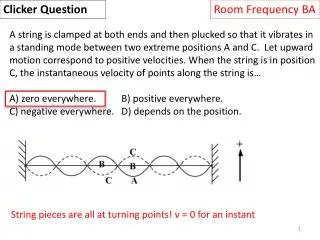
Room Frequency BA Clicker Question Data: cice = 0.50 cal/(goC) , cwater = 1.00 cal/(goC), csteam = 0.48 cal/(goC), L(ice-water)= 80 cal/g , L(water-steam)=539 cal/g Which requires the most energy? A) Raise the temperature of 1 gram of liquid water 50 oC. B) Melt 1 gram of ice at T=0 oC. C) Raise the temperature of 1 gram of ice from 73K to 273K . D) Vaporize 0.5 gram of water at T=100 oC. Q = 50 cal Q = 80 cal Q = 100 cal Q = 269.5 cal Q = mcΔT for no phase change, Q = mL for phase change.
Melting Ice Example How much energy is required to change 1 kg of ice, which is initially at -200°C, into water at 25°C? We will have three different steps: 1) Heat the ice from -200°C to 0°C 2) melt the ice at 0°C 3) then heat water from 0°C to 25°C cice = 2100 J/(kg°C), cwater = 4186 J/(kg°C), and Lice-water = 333 kJ/kg QTotal = (1)(2100)(200) +(1)(333,000)+(1)(4186)(25) = 858 kJ
Room Frequency BA Clicker Question An ice cube is placed in a cup containing some liquid water. The water and ice exchange energy with each other but not with the outside world. After the water and ice come to the same temperature, is it possible the ice could freeze the water rather than the water melt some of the ice? Yes, the ice could freeze the water. No, the water will always melt some of the ice. If the ice is cold enough initially (well below zero), then the heat lost by the liquid to cool it and freeze it could be more than the heat gained by the initial cold ice!