Atomic Binding - Molecular Bonds
Atomic Binding - Molecular Bonds. Atoms are known to come together to form molecules which can be relatively simple (e.g. the hydrogen molecule H 2 ) or very complex (e.g. the DNA molecule).


Atomic Binding - Molecular Bonds
E N D
Presentation Transcript
Atomic Binding - Molecular Bonds • Atoms are known to come together to form molecules which can be relatively simple (e.g. the hydrogen molecule H2) or very complex (e.g. the DNA molecule). • In this part of the course we first identify the interatomic forces which bind atoms to form molecules and, subsequently, study the resulting energy levels (in particular, how they differ from those of the constituent atoms) and spectra of simple molecules. • Finally, we investigate how atoms come together to form solids, the properties of these solids arising from the allowed energy levels and some practical applications of these properties.
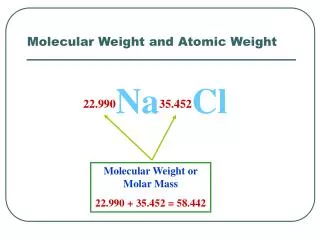
Molecular Bonds - Ionic Bonding • There are two main types of strong molecular bonds namely ionic and covalent bonds. • The sodium chloride (NaCl) molecule provides an example of ionic bonding. Recall that the electronic configuration of the sodium atom is 1s22s22p63s1 andthat of chlorine is1s22s22p63s23p5. • The outermost 3s electron of the sodium atom is weakly bound because it is partly shielded from the nucleus by the inner closed shells (the figure on the next slide shows that the ionisation energy is only 5.1eV). • The 3s electron essentially experiences the effect of a net charge of only +1e because the nucleus has a charge of +11e and there are 10 electrons (charge of -10e) in the inner closed shells.
Ionisation energy (eV) Atomic number, Z Molecular Bonds - Ionic Bonding (ctd.) • The ionisation energy of an atom is the energy required to remove an electron from the atom when it is in the ground state, resulting in the formation of a positive ion.
Molecular Bonds - Ionic Bonding (ctd.) • The chlorine atom has 17 electrons, 12 of which are in closed shells or sub-shells (1s22s22p63s2). These form a spherically symmetric distribution. The remaining five electrons are in 3p states which are not spherically symmetric. • Four of the five 3p electrons are in states whose distributions are in the shape of a doughnut. Since these states are filled, they cannot accommodate the fifth electron. That electron is in another state (in the shape of a dumbbell) which is half filled since it can accept two electrons of opposite spins (refer to the figure on the next slide). • If the weakly bound 1s electron from a sodium atom is in the vicinity of the chlorine atom, it can fill the state. The electron would experience an attraction of +5e because the charge of +17e of the chlorine nucleus is partly shielded by the charge of -12e of electrons in the inner filled shells and sub-shells.
Molecular Bonds - Ionic Bonding (ctd.) Can accommodate 3s electron from sodium atom • The electron is therefore more strongly attracted by the +5e charge of the chlorine atom than the +1e charge of the sodium atom. • It spends most of its time near the chlorine atom which, therefore, acquires a net negative charge in comparison with the sodium atom which acquires a net positive charge. 3s electron Doughnut shaped states Chlorine Sodium
Molecular Bonds - Ionic Bonding (ctd.) • Since the two atoms are oppositely charged, the electrostatic attraction between them forms a bond which holds them together. Such a bond is called an ionic bond because it is due to the electrostatic attraction between two ions of opposite charges. • Many compounds are ionic in nature. Examples are compounds formed from elements in group I (H, Li, Na, K, …) and group (VII) (F, Cl, Br, I, …) of the periodic table. • Ionic bonds that involve more than one outermost electron (valence electron) are also formed from elements of group (II) (Be, Mg, Ca, …) and group (VII) of the periodic table. One example is calcium chloride (CaCl2).
Molecular Bonds - Covalent Bonding • To gain an insight into the formation of covalent bonds, we consider the formation of a hydrogen molecule (H2) from two isolated hydrogen atoms in the ground state. • When the atoms come close to each other, the electron distributions for the two atoms overlap and the two available electrons are shared by the atoms. • This results in two possibilities for the total spin S. Either the spins of the two electrons are parallel in which case S = 1/2 + 1/2 = 1 or The spins are opposite resulting in S = +1/2 + (-1/2) = 0
Molecular Bonds - Covalent Bonding (ctd.) • If the spins are the same (i.e. S =1), the two electrons cannot both be in the lowest energy state for one of the atoms, since this would violate the Pauli exclusion principle (the two electrons would have the same set of quantum numbers). • Therefore when the atoms approach each other, the electron distributions do not overlap. This is shown by the probability distributions in the diagram. • Consequently, the atomic nuclei repel each other and bonding of the two hydrogen atoms does not occur. Electron clouds Probability distribution
Molecular Bonds - Covalent Bonding (ctd.) • If the spins are opposite ( i.e. S = 0),the two electrons have different sets of quantum numbers and can come together (i.e. the electron distributions can overlap). • The electrons, therefore, spend much of their time between the two nuclei. • The resulting attraction of the two nuclei by the electron cloud predominates over electrostatic repulsion of the positive nuclei. • The net attraction of the two atoms holds them together, forming a covalent bond. • Note that in this case the electrons are shared by the two atoms. Electron cloud Probability distribution
Molecular Bonds - Partially-ionic Nature of Covalent Bonds • Oxygen has 8 electrons in the configuration 1s22s22p4. Of the 8 electrons, 4 are in closed shells or subshells. • Electrons from the hydrogen atoms are more attracted to the oxygen nucleus (effective charge of +4e due to shielding effects) than to their own nucleus. • The two H atoms are therefore more positive than the Oxygen atom, which is more negative, due to the imbalance in the electron probability distribution. • This imbalance causes the molecule to be polar and is the basis of its ability to solvate atoms and other molecules, particularly ionic species. • The properties of molecules may be traced to the quantum mechanical nature of their electron distributions.
Molecular Bonds - Other Types of Bonds • In addition to the strong ionic and covalent bond, there are other types of bonds such as the van der Waals bond and the hydrogen bond which are weak bonds. • The van der Waals bond is due to weak electrostatic attractions between molecules. It is important in liquids and solids at room temperature, when thermal excitations are negligible. • The hydrogen bond plays an important part in holding many organic molecules together.
Molecular Bonds - Binding Energy • From the discussion so far, it is clear that two atoms come together to form a molecule because of a net attractive force between the atoms or ions formed. • For the molecule to be stable, the total energy of the molecule must be less than the total energy of the atoms when the are separated. • Energy must, therefore, be supplied to overcome the bond and separate the atoms to infinity. • The required energy is known as the Binding Energy. It is typically 2 to 5eV for ionic and covalent bonds.
Molecular Spectra - Potential Energy Diagrams • Solution of the Schrodinger equation requires knowledge of the potential energy of a system of atoms as a function of the internuclear separations. • For two point charges q1(C) and q2(C), the potential energy U(r) is given by Potential energy term (function of x only in this case) • As the charge separation decreases, U(r) increases for like charges (figure a) because of the repulsive force between them, but decreases for unlike charges (figure b) because the force is attractive.
Molecular Spectra - Potential Energy Diagrams • The figure shows the potential energy of a system of two atoms (e.g. two hydrogen atoms) as a function of the internuclear distance r. • At infinite separation, the force between the atoms is zero, and so is the potential energy. • The potential energy decreases as the atoms approach each other, but for very small separations, the force is repulsive. These are related to changes in the electron distribution between the atomic nuclei. • At a particular separation ro, called the equilibrium separation, the potential energy is a minimum. • The binding energy is roughly equal to the depth of the potential ‘well’. They are not equal because the ground state energy may not lie exactly at the minimum of the potential energy curve.
Molecular Spectra - Potential Energy Diagrams (ctd.) • The total potential energy can be approximated by an expression of the form where A and B are constants related to the attractive and repulsive potentials respectively, and m and n are small integers. U(r) (repulsive) r Ground state energy (attractive)
Molecular Spectra • The energy states of a molecule arise from: (a) rotation of the molecule as a whole (b) vibration of its atoms relative to one another (c) changes in its electronic configuration • The spectra of polyatomic molecules can be very complicated. In this course, the treatment will be confined to diatomic molecules for simplicity. However, the main conclusions will also apply to more complicated molecules.
Molecular Spectra • The spectrum of a molecule carries information about the structure of the molecule. Parameters such as the bond lengths, bond angles and force constants can often be determined from measured spectra. • Different types of molecules have characteristic spectra which can be used for identification. This is done by comparing unknown spectra with ‘fingerprint’ spectra of known molecules. • Solution of the Schrodinger equation for molecules shows that the rotational and vibrational energies of a molecule are quantised. • Transitions between rotational and vibrational energy levels are subject to selection rules (as is the case for atomic transitions), and result in molecular spectra which consist of closely spaced spectral lines. These are known as band spectra.
Quantum number L = 0, 1, 2, 3, ... Molecular Spectra - Rotational States • Consider a diatomic molecule that is rotating bout an axis through its centre of mass (CM) perpendicular to the line joining the atoms. • Its rotational energy Erot is given by where I is the moment of inertia and w is the angular velocity. • The rotational angular momentum of the molecule is equal to Iw. • Solution of the Schrodinger equation shows that the rotational angular momentum Iw is quantised and given by
Molecular Spectra - Rotational States • Consequently, the rotational energy Erot is also quantised and given by L = 0, 1, 2, 3, ..... • Transitions between rotational energy levels are not all allowed. The selection rule is DL = 1 • For a transition between two rotational energy levels with quantum numbers L and L - 1 (remember that DL = 1), the energy DErot of the photon that is emitted or absorbed is L Emission Absorption L - 1 In the above expression, L is the quantum number of the upper state.
Since for a given molecule, the rotational spectral lines are equally spaced in frequency. Molecular Spectra - Rotational Spectra • If f is the frequency of emitted or absorbed radiation, Erot Emission Absorption Or Frequency f Rotational spectrum
where is called the reduced mass. Molecular Spectra - Rotational States • The moment of inertia I of the diatomic molecule shown in the figure is given by • Since the molecule rotates about an axis through its centre of mass (1) (2) • From equations (1) and (2), noting that r = r1+r2, the moment of inertia can be written as
Rotational Spectra- Example Example The rotational transition from L = 0 to L=1 of the CO (carbon monoxide) molecule has an absorption frequency of 1.15x1011Hz. The mass of 12C is 1.99x10-26 kg and that of 16O is 2.66x1-26 kg. (a) What is the moment of inertia of the CO molecule? (b) What is the CO bond length? (c) What is the wavelength of the emitted photon for the transition from L = 4 to L = 3?
Rotational Spectra- Example The frequency f is given by f = 1.15x1011 Hz, h = 6.626x10-34 Js, L = 1 (for upper state) (a) (b) (c) Emission is in the microwave spectral region (l ~ 0.1mm - 10mm)
Molecular Spectra - Vibrational States • We have seen that molecular energy states can result from rotation of a molecule as a whole. • Molecular energy states can also result from vibrations of the atoms in a molecule relative to one another. • Near the equilibrium position ro, the potential energy U can be approximated by that of a harmonic oscillator. Displacement Uo • The restoring force F is given by (Hooke’s law) • For small displacements about ro, the restoring force is proportional to the displacement and the molecule undergoes simple harmonic motion about ro.
Molecular Spectra - Vibrational States • For a simple harmonic oscillator, the classical frequency f of oscillation is given by k is the stiffness constant m is the reduced mass • Solution of the Schrodinger equation for the simple harmonic oscillator potential shows that the oscillator energy Evib is quantised. Vibrational quantum number v = 0, 1, 2, 3, .... f is the frequency • Note that the lowest vibrational energy ( for v = 0) is not zero (as is the case for rotation) but hf/2. This is known as the zero point energy. • Also note that the energy levels are equally spaced. Energy spacing is hf.
Molecular Spectra - Vibrational States • Vibrational transitions are subject to the following selection rule: • The selection rule shows that allowed vibrational transitions can only occur between adjacent vibrational energy levels. • In the simple harmonic approximation, the energy DEof emitted or absorbed photon is given by Dv = 1 • Transition energies are 10 to 100 times those for rotational and wavelengths are in the infrared spectral region (l ~ 1mm to 100mm)
Molecular Spectra - Vibrational States • Example The hydrogen molecule emits infrared radiation of wavelength of approximately 2.3mm. (a) What is the energy separation (in eV) of adjacent vibrational levels? (b) What is the energy (in eV) of the lowest vibrational state? (a) (b) For lowest vibrational state, v = 0
Molecular Spectra - Anharmonic Oscillators • For large displacements ( larger than ~ 10% of bond length), molecular vibrations are not simple harmonic in nature. • The shape of the potential energy curve is more complicated as shown by the solid line in the top figure (the dotted line shows the simple harmonic oscillator approximation). Displacement v • The vibrational energy levels are then not equally spaced, and transitions frequencies and wavelengths are different from those of the simple harmonic oscillator approximation. 5 4 Energy 3 Energy levels for large displacements 2 1 0 Internuclear distance
6 6 5 5 4 4 3 3 2 2 1 1 0 0 Molecular Spectra - Rotational and Vibrational transitions • Molecules can rotate and vibrate simultaneously. Transitions between rotational and vibrational energy levels of a molecule are subject to the following selection rule: DL = +1 DL = -1 DL = 1 and Dv = 1 L • The figure shows some transitions between the rotational-vibrational energy levels of a diatomic molecule and the resulting spectrum. • Note that there is a gap in the spectrum corresponding to DL = 0. v = 1 v = 0 Energy
Molecular Spectra - Rotational and Vibrational transitions • The figure shows the absorption spectrum of the HCl molecule. • Each line is made up of two peaks because chlorine has two isotopes which have different masses and, therefore, different moments of inertia.








![Atomic Bonds [aka chemical bonds]](https://cdn1.slideserve.com/3075537/atomic-bonds-aka-chemical-bonds-dt.jpg)