Blackbody Radiation
530 likes | 1.33k Vues
Blackbody Radiation. • All bodies emit EM radiation because of their temperature (thermal radiaiton) but they are not usually in equilibrium. (but radiation emitted: thermal radiation+reflected) • However consider an opaque enclosure with walls at constant temperature.

Blackbody Radiation
E N D
Presentation Transcript
Blackbody Radiation • All bodies emit EM radiation because of their temperature (thermal radiaiton) but they are not usually in equilibrium. (but radiation emitted: thermal radiation+reflected) • However consider an opaque enclosure with walls at constant temperature. Radiation and walls reach thermal equilibrium. Under these conditions the radiation has quite definite properties. •How can we study this radiation? If we cut a tiny hole in the enclosure it will not disturb the eqbm. And the emitted radiation will have the same properties as the Cavity Radiation. •Emitted spectrum = spectrum from a perfect black body at the same T as the enclosure. For this reason cavity radiation is also called black-body radiation.
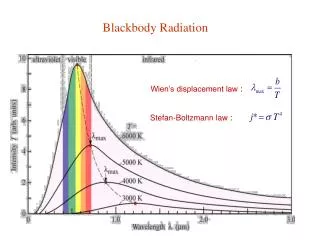
Blackbody Radiation • Experimental results: • Stefan-Boltzmann law • PA = T4 , where • = 5.67 x 10 -8 Wm -2 K -4 PA is the power emitted per unit surface area. Wien’s displacement law λmax.T = const = 2.9 x 10 -3 mK
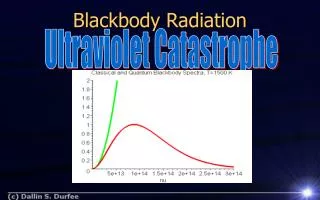
Origin of Blackbody Radiation The origin of the em. waves in the cavity must relate to the thermal motion of charged constituents of atoms in the material (we know they are electrons) Basic idea = radiation incident on a surface is reflected because it is quickly absorbed and re-emitted. Rayleigh and Jeans worked out the spectral shape (classical theory). Each wavelength is emitted by atoms oscillating at a frequency = c/λ. The predicted spectrum is then the product of two functions - N() the number of oscillators with freq. , which is proportional to 2 and The average energy of an oscillator with freq. , which is equal to kT Result S prop. to 2, which is infinite at large , small λ Ultraviolet catastrophe
Rayleigh-Jeans Formula The assumptions made by Rayleigh and Jeans lead to the spectrum for a blackbody marked “Classical Theory” below. Note:-Here the spectrum is plotted as a function of frequency.
Rayleigh-Jeans Formula Here it is plotted as the more familiar function of Wavelength.
Physical Picture of Planck’s quantum hypothesis ●Assume atomic oscillators behave like simple harmonic oscillators with V = ½ kx2 parabola
Sun-Yellow Red • Spectrum of Blackbody Radiation • as a function of wavelength. • Energy emitted by four blackbodies with • equal surface areas. • Note that they are plotted on log-log scale. • Area is proportional to total power per unit surface • area (PA) • Stefan-Boltzmann Law: PA = .T4 • PA is in Wm-2 and = 5.67 x 10 -8Wm-2 K-4
Blackbody Radiation • General question:-What is the spectrum of EM radiation emitted by an object of arbitrary temperature T in thermal equilibrum. We assume that this “blackbody” reflects no radiation at any •Max Planck showed that the spectrum is given by ud = 8hc -5.d [exp(hc/kT) - 1] ud is the energy density =energy/unitvolume • Although no perfect blackbody exists solids and stars follow Planck’s Law very closely. Note that picture is on log-log scale.
Wien’s Displacement Law • Doubling T increases P by 16 since PA = .T4 • Note that maximum wavelength max shifts with T.This can be quantified in Wien’s Displacement Law. max.T = const. = 2.9 x 10-3 mK • This quantifies the observation that an object changes colour with Temperature e.g.At room temp. spectrum peaks in infra-red. • Very important since it allows us to obtain a measure of the SURFACE TEMPERATURE of a star from max.For the Sun max is in blue witha lot of radiation in red so it looks yellow.For stars with T = 3000k max is in infrared but significant amount in red.Red Giants are at this T.
Planck’s Solution ●Planck decided that the problem lay in the assumption that the average energy of the oscillators is a constant and not in the number of modes of oscillation. ●He looked for a way of reducing the number of high frequency oscillators in the cavity. In other words there is too much radiation at small λ. ●He suggested the idea that an oscillating atom can absorb or re-emit energy only in discrete bundles or quanta. ●If the energy of the quanta are proportional to the frequency() then as becomes large then becomes large. Since no standing wave can have E>kT, no standing wave can exist with an energy quantum larger than kT. This effectively limits the high frequency (small wavelength) radiant intensity and removes the “Ultraviolet catastrophe”. [Max Planck Nobel Prize 1918
Planck’s Solution ● Planck had two new ideas. 1)he assumed that the energy allowed per oscillator is not continuous. In other words the oscillators can only have certain discrete values of energy. Energy is quantised. 2)He assumed that the gaps between allowed values of energy are greater for high frequency oscillators. He made the simple guess that E = nh, where h = const., n = 1,2,3,4,----- and the energy of each quantum is h. The average energy per osc. then tends to zero as increases. •Max Planck showed that the spectrum is given by ud = 8hc -5.d Wm-2 [exp(hc/kT) - 1] This fits perfectly if h = 6.63 x 10 -34 Js Note:-Planck was slow to accept this-like others.
The Photoelectric Effect • Many people, including Planck, were very sceptical about his solution to the Black-body radiation problem.The drawback was the idea of quantisation of the energies of the oscillators. • The thing which convinced them was Einstein’s explanation of the Photoelectric Effect. • The phenomenon was first observed by Hertz in 1887. He noticed that the air in a spark gap became a better conductor when illuminated by UV light. • It was then studied in detail by Hallwachs, Lenard, Millikan and others. Basically they showed that electrons are emitted from a metal surface when illuminated by light of sufficiently high frequency. In other words there is a threshold in frequency before anything happens. •It needs a systematic study to highlight the important features.
Photoelectric effect-Results to be explained • The number of electrons ejected per unit time • is proportional to the intensity of the Light. • 2)The electrons are emitted with velocities up to a • maximum velocity(VSTOP < 0 and # 0) • 3)The maximum kinetic energy does not depend on • intensity. • 4)There is a THRESHOLD FREQUENCY0 such • that there is no emission for < 0 • The threshold frequency depends on the metal. • 5)There is no measurable delay between the light • striking the metal and the emission of the electrons • no matter the incident light intensity,i.e.no matter • how low the intensity.
Wave Theory and Photoelectric Effect. • Wave theory would suggest that the maximum kinetic energy should depend on the intensity of light. •The wave theory predicts no threshold effect. It would be simply a matter of collecting enough energy. •A measurable time delay would be expected. Enough energy has to be accumulated before an electron can be emitted. So the Wave Theory has failed but Einstein had a simple theory based on quantisation.
Einstein’s Theory of Photoelectric Effect. •Einstein proposed that light (EM radiation) consists of particle-like packets of energy called photons •Each photon carries an energy E = h where h = Planck’s constant, = wave frequency •This extends Planck’s ideas regarding emission and absorption of radiation so that they apply when the radiation is transmitted. •The emission of electrons is caused by single photons being completely absorbed by individual electrons. • •The energy is called the Work Function.the maximum kinetic energy for electrons is then Kmax = h - Note:-We can have K < Kmax for tightly bound electrons or because of collisons during emission. Free electrons E Bound Electrons
Photoelectric Effect-Einstein’s Hypothesis ● Einstein proposed that radiation is quantised not just in absorption and emission but in all situations. •The energy is called the Work Function •The max. kinetic energy for electrons is Kmax. = h - we can have K < Kmax for tightly bound electrons or collisions during the emission of the electrons •Note that Kmax = VSTOP x e where e = charge on electron •So VSTOP plotted vs. has slope h/e and we can measure h and compare it with value from BB radiation formula. •They agree with h = 6.626 x 10-34 Js = 4.136 x 10-15 eV.s •The Einstein model accounts for all 5 experimental features of the photoelectric effect.
Photoelectric effect-Results to be explained 1)The no.of electrons ejected per unit time is propnl. to the intensity of the Light. [ It is proportional to the no. of photons] 2)The electrons are emitted with velocities up to a maximum velocity(VSTOP < 0 and # 0) [Kmax = h - ] 3)The maximum kinetic energy does not depend on intensity. [ It depends instead on the energies of the photons] 4)There is a THRESHOLD FREQUENCY0 such that there is no emission for < 0 The threshold frequency depends on the metal. [ if h < then we cannot release an electron] 5)There is no measurable delay between the light striking the metal and the emission of the electrons no matter the incident light intensity,i.e.no matter how low the intensity.
The Compton Effect •Arthur Compton(1923). His experiments showed definite particle-like(photon) behaviour for X-rays. •Simply the result is () function of (0) ' = + •Compton scattering occurs in addition to the classical process of Thomson scattering where () = (0) and we have absorption and re-radiation. The Compton shift has a definite ang. dependence. •It does not depend on the precise material used as the scatterer. •This suggests that the photons collide with something which is in all materials,namely electrons.
Compton Effect •Compare X-ray photon energy with electron energies -Binding energies are few eV •X-ray photon with = 0.07nm,the wavelength used by Compton. E = h = hc/ =6.63.10-34.3.108 0.07.10-9 1.6.10-19 = 17.7 keV »electron binding energy •Consider a collision between a photon and a free unbound electron at rest. For a photon, E = pc [A photon must have m0 =0 since =1 ] •We can calculate the results for such a collision assumimg 1)Conservation of momentum 2)Conservation of mass-energy 3)Energy-momentum relationship
Compton Scattering •Collision can be analysed in two dimensional lab. Frame •Photon momentum is given by p = E/c = h/c •We can resolve momentum in the horizontal(pL) and transverse(pT) directions pL: h/c = h'/c x cos +pecos pT: 0 = h'/c x sin =pesin E: h + m0c2 = h' + sqrt(pe2c2 + m02c4 ) What we want is an expression for the final frequency ' as a function of the initial frequency and the scattering angle and must eliminate the unknowns and pe
Compton Scattering Schematic of Compton Scattering process. We can also solve for the energy of the scattered Photon. h / = h0 [ 1 + h0/ m0c2( 1 – cos θ )] • When θ = 1800 this gives an energy for the • scattered photon of ~ m0c2/2if the energy of the • incident photon is much larger than the rest mass • of the electron. • b) When θ = 900 this gives an energy of ≈ m0c2 for • the scattered photon when the incident photon • energy is large.
Photons The ideas of Planck and Einstein and the experiments of Compton seem to show conclusively that light is composed of photons. However, earlier , we had decided with the 19th C. physicists that light is a waveform since it describes very well the phenomena of interference and diffraction. How can we reconcile the two. Let us first look at what happens as we allow only a tiny amount of light through an optical spectrum. This gives an insight into the situation.
1AMQ, Part II Quantum Mechanics4 Lectures Wave Particle Duality, the Heisenberg Uncertainty Principle, de Broglie (wavelength) hypothesis Schrodinger Wave Equation. Wavefunctions and the Free Particle. Electrons in a box, energy quantisation. • K.Krane, Modern Physics, Chapters 4 and 5 • Eisberg and Resnick, Quantum Physics, • Chapters 3 (and 5 & 6)
Wave-Particle Duality. Light behaves like: (a) a wave (diffraction, interference) and/or (b) a stream of massless particles orphotons, (black body spectrum, photoelectric effect, Compton scattering). Consider diffraction from a slit, one photon at a time. The experiment shows that individual particles of light gradually build up the diffraction pattern predicted by the classical wave theory. The wave pattern describes the probability of detecting a photon at that point. The wave pattern measures the probability detecting a photon at that point.
Individual photons unpredictable in detail, BUT their average behaviour is predictable! At any point P in the interference pattern, the intensity gives the probability: i.e. I(P) is prop. to prob. that a photon hits pos. P. For waves, the intensity = (amplitude)2 and thus the wave and particle pictures are connected, Probability a (amplitude of wave)2 This is called the Born Interpretation (Max Born, Nobel Prize, 1954). The question then arises, if waves can act like particles, is the reverse true ? The de Broglie Hypothesis Louis de Broglie (for his PhD thesis!) postulated that in some phenomena, particles can act like waves! (Nobel Prize, 1929)
He guessed that for particles, the momentum, p, could be related to a wavelength, l , by p = h/l where l is the de Broglie wavelength. Note this is consistent with the momentum for photons, ie p = E/c = hn/nl = h/l This predicts, for example, that electrons should show diffraction effects (like photons), if they pass through a slit with width ~ ldB. Calculating the de Broglie Wavelength. Example. e-s in a TV Ke-=20 keV, me=9.1x10-31kg, gives ldB=0.009nm. (Note that the typical atomic spacing is ~0.05nm). ldB=0.05nm corresponds to Ke-=601 eV for electrons. [from Ke=p2/2me=(1/2m) . (h/l)2] ie. e-s accelerated across 600 V should be diffracted exactly like x-rays with l=0.05nm.
X-ray diffraction pattern Electron diffraction pattern
Note how (2 slit) e- interference pattern builds up with increasing number of e-s. Electron and neutron diffraction are widely used in crystallography studies.
The y-position for any transmitted photon is specified to an accuracy (uncertainty) D y=a. Making the slit smaller (ie. reducing D y) increases the uncertainty in the angle, Dq.
Thus, DyDpy~ h (for single slit diffraction). This relates to the physical limits we can put on determining either the position or momentum of the individual photons as they pass through the slit. These are a subset of the Heisenberg Uncertainty Principle. In general, if p = momentum, t = time and E= energy, h = Planck’s constant ,then, The division by 2p comes about due to the use of wave number (k) rather than wavelength (l=2p/k) in the definition of momentum (see Krane p114).
Basis for Schrodinger’s equation • Light can be described - as a wave - as a stream of particles }in some circumstances • Following De Broglie we can say that Matter also has this dual wave-particle character. • Probability | Amplitude of wave | 2 - the Born Interpretation. •Uncertainty Principle - Heisenberg p.x h/2 E.t h/2
The Schrödinger Equation. For particles, forces change momentum (p=mv) and each force has an associated potential energy. Newton’s 2nd Law relates the potential to the change in momentum via the relations Solving the Schrodinger equation specifies Y (x,t) completely, except for a constant, ie. if Y is a solution, so is A xY . From the Born interpretation we have ||2.dx as the probability of finding the particle at position x.Since the particle must be somewhere the integral of this quantity from - to + must equal 1.0.This allows us to normalise the wavefn. We will solve this equation for the wavefunction with an amplitude as a function of position and time t
Time Independent Schrodinger Equation In many cases, (eg. Coulomb potential in H atom), V=V(x), ie. not time dependent, then where y(x) satisifies • Properties of the T.I.S.E. • Applies when V=V(x), ie. V is independent of • time.Eg. an e- wave acted on by a fixed nucleus, • but NOT an atom in an oscillating magnetic field. • Applies only in the non-relativistic limit • (ie. assumes K=(1/2)mv2=p2/2m). • Assumes p=h/l and E=hn. • Is linear iny, ie no terms like y2, y3 etc. Laws • of superposition are valid. If ya and yb are • two possible solutions, then y=caya+cbyb • is a solution for all constants ca andcb.