Understanding Phase Transitions and Thermodynamic Reactions in Chemistry
This detailed guide explores key concepts in thermodynamics, focusing on phase transitions of water (ice, liquid, steam) at different temperatures. It examines the calculations of enthalpy changes (ΔH) during reactions, specifically highlighting the Thermite reaction and Hess's Law. The guide delves into heat transfer involving specific heat capacities for ice, water, and steam, as well as calculations for bond energies in chemical reactions. Utilize this information to strengthen your understanding of thermodynamic principles and their applications in chemical reactions.


Understanding Phase Transitions and Thermodynamic Reactions in Chemistry
E N D
Presentation Transcript
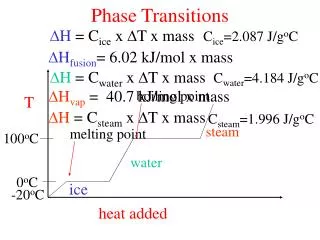
melting point boiling point 100oC 0oC Phase Transitions H = Cice x T x mass Cice=2.087 J/goC Hfusion= 6.02 kJ/mol x mass H = Cwater x T x mass Cwater=4.184 J/goC Hvap = 40.7 kJ/mol x mass T H = Csteam x T x mass Csteam=1.996 J/goC steam water ice -20oC heat added
Thermite reaction Al2O3(s) + 2Fe(l) 2Al(s) +Fe2O3(s) H is an Exothermic extensive, State function Hess’ Law
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) 2Al(s) + 3/2 O2(g) Al2O3(s) H = -1676 kJ/mol _______________________________ __________ Fe2O3(s) 2Fe(s) + 3/2 O2(g) 2 Fe(s) + 3/2 O2(g) Fe2O3(s) H= - 822 kJ/mol + Al2O3(s) + 2Fe(s) -854 kJ/mol 2Al(s) + Fe2O3(s) 2 ( ) +15 kJ/mol 2 2 Fe(s) Fe(l) _______________________________ __________ 2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) Hrxn = -824 kJ/mol
Hess’ Law • Always end up with exactly the same reactants and products • If you reverse a reaction, reverse the sign of H • If you change the stoichiometry, change H
Heats of formation, Hof H = heat lost or gained by a reaction “o” = standard conditions: all solutes 1M all gases 1 atm “f” = formation reaction: 1mol product from elements in standard states for elements in standard states, Hof= 0
Write the equation for which Hrxn = Hof Hof for NH2CH2COOH from elements 1 mol product, in their standard states NH2CH2COOH + O2 N2 + H2 + Cgr 2 1/2 5/2
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) products reactants elements Hof 2 Al(s) Al2O3(s) 2 Al(s) 2 Fe(s) Fe2O3 2 Fe (l) 3/2 O2(g) Hof Al2O3(s) + 2 HofFe (l) Al(s) Fe2O3
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) products reactants elements Hof 2 Al(s) Al2O3(s) 2 Al(s) 2 Fe(s) Fe2O3 2 Fe (l) 3/2 O2(g) Hof Al2O3(s) + 2 HofFe (l) - Hof - Hof Fe2O3 Al(s) - Hrxn = nHofproducts nHofreactants
2Al(s) +Fe2O3(s) Al2O3(s) + 2Fe(l) - nHofreactants Hrxn = nHofproducts [HofAl2O3(s) + 2 HofFe(l)] Hrxn= - [HofFe2O3(s) + HofAl(s)] 2 [(-1676) + (15)] 2 - [(-822) + 0]kJ Hrxn = = -824 kJ
Bond Energies chemical reactions = bond breakage and bond formation bond energies positive energy required to break bond bond breakage a) endothermic b) exothermic (raise P.E.) bond formation exothermic (lower P.E.)
Bond energies CH4 (g) + 2O2 (g) CO2 (g) + 2H2O (g) Hrxn= bonds broken C-H 413 kJ O=O 495 kJ C=O 799 kJ O-H 467 kJ - bonds formed Hrxn= 4 [ (C-H) + (O=O)] 2 - [ (C=O) 2 + (O-H)] 4 = -824 kJ Hrxn= Hof products - Hof reactants =- 802 kJ