Cohort study
Cohort study. Dr K N Prasad MD., DNB Community Medicine. Aim of Epidemiological studies. To determine distribution of disease To examine determinants of a disease To judge whether a given exposure causes or prevents disease. Epidemiological study designs. Descriptive studies

Cohort study
E N D
Presentation Transcript
Cohort study Dr K N Prasad MD., DNB Community Medicine
Aim of Epidemiological studies • To determine distribution of disease • To examine determinants of a disease • To judge whether a given exposure causes or prevents disease
Epidemiological study designs • Descriptive studies • Populations • Correlated studies • Individuals • E.g. case-series, case reports, cross-sectional surveys • Analytical studies • Observational studies • Case-control studies • Cohort studies • Experimental studies • Intervention studies after randomise exposure • Clinical trials
Case-control study Exposed Cases Non-exposed Study Population Exposed Controls Non-exposed
Disease + Exposed Disease - Study population Disease + Non-exposed Disease - Cohort study / Follow-up study
General considerations A cohort : A group of persons, identified at one point in time, who march off together into the future under the watchful eye of an investigator. A cohort study: A group of persons is defined, certain characteristics about each individual are recorded, and they are then followed up in such a way that new events (such as disease and death) or other changes in their characteristics are detected.
Cohort Study Longitudinal study, Follow-up study, prospective study Definition: An analytical epidemiological study in which two or more groups of people according to the extent of exposure (e.g. exposed and unexposed) are compared with respect to outcome or disease incidence Most reliable for showing an association between a suspected risk factor and subsequent disease
Features of cohort study • Cohorts must be free from the disease under study • Both the groups should be equally susceptible to disease under study • Diagnostic and eligibility criteria of the disease must be defined beforehand
Cohort study • Exposed and non exposed individuals are followed over time to determine whether they experience the outcome of interest. • Examples of exposure : • Medication use, Environmental factors, condition, Procedure • Examples of outcome: • Disease. Death, etc.
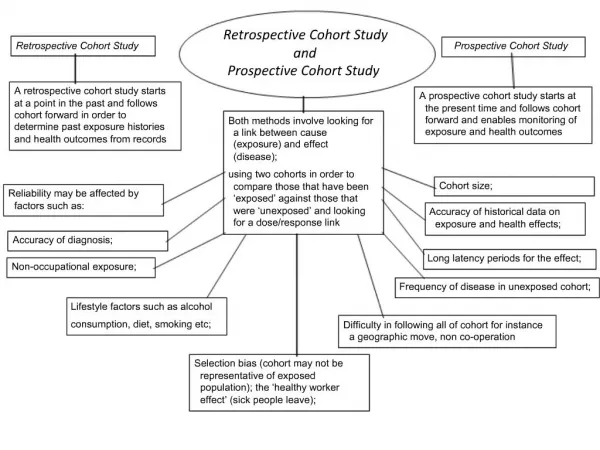
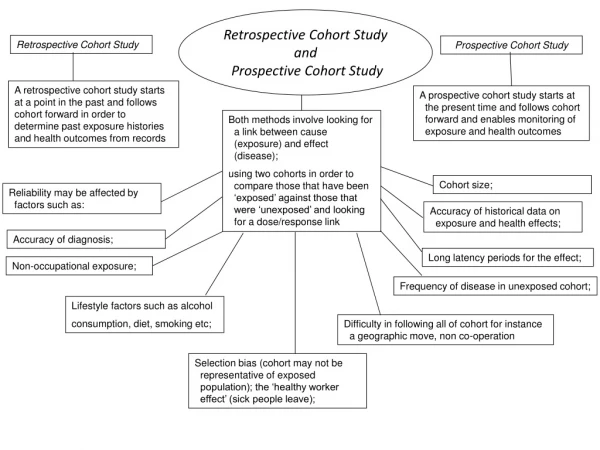
Cohort studies • Retrospective • Exposure Disease • Yes ? • No ? • Prospective • Exposure Disease • Yes ? • No ? • Ambidirectional
Timing of cohort studies • Retrospective: both exposure and disease have occurred at start of study Exposure------------------------Disease *Study starts
Timing of cohort studies • Prospective: exposure has (probably) occurred, disease has not occurred Exposure----------------------Disease *Study starts • Ambi-directional: elements of both
Elements of cohort study • Selection of study subjects( cohorts) • Selection of comparison group • Obtaining data on exposure • Follow up • Analysis
Selection of the Exposed Population • Sample of the general population: • Geographically area, special age groups, birth cohorts (Framingham Study) • A group that is easy to identify: • Nurses health study • Special population (often occupational epidemiology): • Rare and special exposure • Permits the evaluation of rare outcomes
Selection of the Comparison Population • Internal Control Group • Exposed and non-exposed in the same Study population (Framingham study, Nurses health study) • Minimise the differences between exposed and non-exposed • External Control Group • Chosen in another group, another cohort (Occupational epidemiology: Asbestosis vs. cotton workers) • The General Population
Selection of comparison group • Internal comparison group: according to the degrees or levels of exposure • Smokers, BP, Alcohol, diet etc. • External comparison • Similar in all respects without any exposure • Comparison with general population rates • Outcomes are compared with the similar outcome rates in the general population
Sources of exposure information: Pre-existing records - inexpensive, data recorded before disease occurrence but level of detail may be inadequate. Records may be missing, / usually don't contain information on confounders
Sources of exposure information: • Questionnaires, interviews: • good for information not routinely recorded but have potential for recall bias • Direct physical exams, tests, environmental monitoring may be needed to ascertain certain exposures.
Follow up • Regular follow up of all participants • Periodic medical examination of each member • Reviewing physician and hospital records • Routine surveillance of death records • Mailed questionnaires, telephone call, periodic home visits
Sources of outcome information: • Death certificates • Physician, hospital, health plan records • Questionnaires (verify by records) • Medical examinations
Analysis in cohort study • Incidence of disease among exposed and non exposed • Relative risk estimation • Attributable risk estimation
Table for analysis cohort study Incidence of disease among exposed = a / a+b Incidence of disease among non exposed = c / c+d P value should be <0.05
Relative risk Relative risk is calculated as Incidence of disease among exposed Incidence of disease among non exposed
Relative risk (Risk ratio) • Quantifies magnitude of the association between exposure and disease • Varies from 0 to infinity • RR<1: exposure decreases the risk for disease • RR=1: no association • RR>1: exposure is a risk factor for disease; increases risk for disease • Example: • RR=2.0 can be interpreted as two fold increase in risk
Attributable risks • Also known as risk difference • It is the difference in incidence rates of disease between exposed group and non exposed group. • It suggests the amount of disease that might be eliminated if the risk factor could eliminated or controlled. Incidence of disease among exposed - incidence of disease among non exposed ------------------------------------------------------- x 100 Incidence of disease among non exposed ex. AR is 90%. Interpretation-
Bias in Cohort study • Selection bias - less of a problem than case control studies • Information bias/misclassification • Degree of accuracy of classification of exposure, confounders and disease status • Loss to follow-up (affects validity) • Non response (limits generalisability, not validity) • Confounding
Limitations Loss to follow-up Misclassification of disease or exposure status logistically challenging – especially for prospective design Hard to study rare diseases Changes over time in staff/methods Little control over nature and quality of data in retrospective designs Strengths Can establish time order Can obtain incidence rates Can study more than one disease or outcome Minimizes bias in ascertainment of exposure status and covariates – especially if collecting data prospectively Efficient for rare exposures No controls, so no bias in control selection Cohort study
Cohort study Rare exposure Examine multiple effects of a single exposure Minimizes bias in the in exposure determination Direct measurements of incidence of the disease Validity can be affected by losses to follow-up Case-control study Quick, inexpensive Well-suited to the evaluation of diseases with long latency period Rare diseases Examine multiple etiologic factors for a single disease Selection Bias and recall bias
Key points in Cohort study • Presence or absence of risk factor is determined before outcome occurs • Identify cohort (s). • Measure exposure and outcome variables • Follow for development of outcomes • Estimate incidence rates, RR and AR, if possible population AR.
Thought for the day Motivation is what gets you started. Habit is what keeps you going. -Jim Ryun
Strengths of Cohort Studies • Efficient for rare exposures, diseases with long induction and latent period • Can evaluate multiple effects of an exposure • If prospective, good information on exposures, less vulnerable to bias, and clear temporal relationship between exposure and disease
Weaknesses of Cohort Studies • Inefficient for rare outcomes • If retrospective, poor information on exposure and other key variables, more vulnerable to bias • If prospective, expensive and time consuming, inefficient for diseases with long induction and latent period • Keep these strengths and weaknesses in mind for comparison with case-control studies
Cohort study • Example of cohort studies • The association between statin use and prostate cancer risk • All men 45+ years enrolled in GHC for at least 2 years during 1990-2004 • Exposure is statin use, which may change over 14 years • Follow 14 years until develop prostate cancer, die, or disenroll from GHC • Each subject will contribute person-time to follow-up Survival analysis to account for time varying exposure, adjust for other risk factors, & account • for censoring • Prostate cancer • No cancer • Statin user • Nonuser • 14 years • Prostate cancer • No cancer
Prospective vs. retrospective Cohort Studies • Prospective Cohort Studies • Time consuming, expensive • More valid information on exposure • Measurements on potential confounders • Retrospective Cohort Studies • Quick, cheap • Appropriate to examine outcome with long latency periods • Admission to exposure data • Difficult to obtain information of exposure • Risk of confounding
Analysis in Cohort study Exposed and non-exposed individuals are followed over time to determine whether they experience the outcome of interest Examples of exposure: Environmental factor, condition, procedure Examples of outcome: Disease, death, costs