Overview of Cell Theory and Chemical Bonding Principles
This review covers the fundamental concepts of cell theory, highlighting four main points: the cell as a basic structural and functional unit of life, the derivation of cells from existing cells, and the interdependence of cells in multicellular organisms. Key scientists such as Robert Hooke, Anton van Leeuwenhoek, John Dalton, and Louis Pasteur are noted for their significant contributions to cell biology. Additionally, the review explores essential chemical bonding concepts, including covalent, ionic, and hydrogen bonds, as well as the role of electrolytes in facilitating electrical currents in biological systems.

Overview of Cell Theory and Chemical Bonding Principles
E N D
Presentation Transcript
Cell Theory and Bonding Review
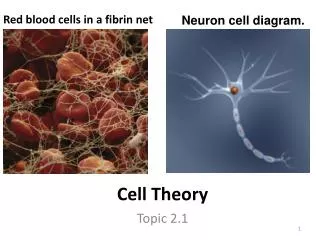
Cell Theory • 4 main points: • Cell is the basic structural unit of living things. • Cell is the basic functional unit of living things. • All cells are derived from previously existing cells. • In a multicellular organism, the activity of the entire organism depends on the total activity of its independent cells.
Key Scientists • Robert Hooke (1665) • First recorded cell observations
Key Scientists • Anton von Leeuwenhoek (1674) • First recorded living cell observations • Animalcules, and which we now refer to as micro-organisms. He was also the first to record microscopic observations of muscle fibers, bacteria, spermatozoa and blood flow in capillaries.
Key Scientists • John Dalton (1808) • Atomic Theory • Could this be true for living things as well?
Key Scientists • Louis Pasteur brought the third point to light in 1864 • Cells are derived from pre-existing cells
Chemical Bonding • Matter takes up space and has mass. • Atoms • Protons, neutrons and electrons • Covalent Bond • Two non metals • Stability • Double or Triple Bonds • Polar covalent bonds
Chemical Bonding • Hydrogen Bonds • Water • Electrostatic attraction • Ionic Bonds • Cation and Anion join • Electrolytes – ions dissolved in water that enable water to carry an electrical current