Matter: Elements, Compounds, and Mixtures
E N D
Presentation Transcript
Let’s start small and work up from there. • Atom – the smallest unit of an element that maintains the properties of the element • The can be physically mixed or chemically joined to make up other forms of matter
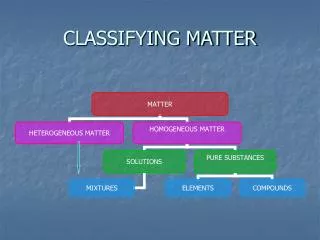
Pure Substance • A sample of matter, either a single element or a single compound, that has definite chemical and physical properties Figure 14, Page 22
Physical Properties • Characteristic of a substance that does not involve a chemical change • Examples: Density, color, hardness • Does this test change the chemical make-up of the material? • No
Chemical Property • Characteristic of matter that describes a substance’s ability to participate in chemical reactions • Example: Oxidation, flammability, and combustion • Does this test change the chemical make-up of the material? • Yes
Elements • A pure substance that contains only one kind of atom • It cannot be separated or broken down into simpler substances by chemical means. • All atoms of the same element have the same atomic number • Elements may bond in different ratios with itself. These are called allotropes.
Compounds • A pure substance that is made up of two or more different elements joined by chemical bonds. • They are represented by formulas
Molecules • The smallest unit of a substance that keeps all of the physical and chemical properties of that substance. • It can consist of one atom or two or more atoms bonded together • In other words, molecules can be of the same element or of the same compound only
All of the following are molecules, but are they compounds or elements? Element • Br2 • H2O • O2 • O3 • C12H22O12 Compound Element Element Compound
Mixtures • A combination of two or more substances that are not chemically combined. • Examples are air, ice tea, and even cake batter • The proportions of the substances can vary
Mixtures can be homogeneous or heterogeneous • Homogeneous mixtures have a uniform structure or composition throughout • Because of the even distribution, any two samples taken from the same homogenous mixture will have the same proportions of ingredients • Examples: Gasoline, air, and syrup
Mixtures can be homogeneous or heterogeneous • Heterogeneous mixtures are not evenly mixed. • Different regions of the same heterogeneous mixture will have different proportions • Examples: Pulpy Orange Juice, chocolate chip cookie dough, and granite.
Mixtures verses Compounds, How can you tell which is which? • Compounds are two or more elements that are chemically bonded together. • Mixtures are two or more substances physically mixed together, but not chemically joined
Therefore • Properties of a mixture reflect the properties of the substances mixed in it; compounds have new properties of their own. • Compounds have definite composition; mixtures composition can vary from mixture to mixture of the same substances
In other words, • You can have a mixture of hydrogen gas and oxygen gas in various concentrations, but the compound water, H2O, will always have 2 hydrogen atoms bonded to one oxygen atom.
Classify each of the following as an element, compound, homogeneous mixture, or a heterogeneous mixture • Iron (III) Oxide (Fe2O3) • 24 Karat gold • Sugar Water • Ozone • Fizzy Soda • Flat Soda Compound Element Homogeneous Mix Element Heterogeneous Mix Homogeneous Mix
Separating Mixtures Since mixtures are just physically combined, they can be separated.
Separating Mixtures • Some Methods include: • Filtering – separation of a mixture’s components through differences in particle size • Decanting – a fancy term for separating two components by pouring • Distillation – used to separate two liquids based on their differences in boiling points • Magnetism – used to separate magnetic substances • Evaporation – removing a liquid to leave a solid behind • Centrifuge – separates substances of different densities using a fast rotational motion • Chromatography – Separates two substances by using a mobile phase and a stationary phase