Chemiluminescence Detection: NO2 Content Measurement in Sample Gas
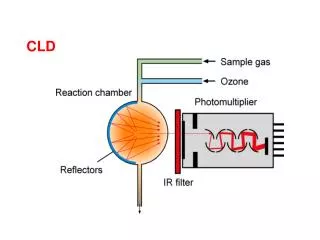
Chemiluminescence method using ozone-mixed dry air to measure NO2 content in sample gas. Reaction chamber converts NO to NO2, detected via infrared radiation. Quenching effects addressed through reduced pressure and optimized wavelength range.


Chemiluminescence Detection: NO2 Content Measurement in Sample Gas
E N D
Presentation Transcript
With dry air produced ozone is mixed with sample gas. • The mixture enters the reaction chamber, while the NO of the sample gas reacts to NO2 : • NO + O3 NO2 + O2 + h • A proportion of the reaction energy is emitted as infrared radiation(h). • The infrared radiation is detected, enhanced and transformed into the detector signal by the photomultiplier. • The NO2 of the sample gas is converted into NO by the integrated converter and then measured in a separate reaction chamber. • The difference between both measurements corresponds to the NO2 content of the sample gas.
CLD - Measuring Principle The chemiluminescence is disturbed by “Quenching” effects. They are caused by absorption of the emitted radiation by other components and by radiation-free energy loss due to collision processes. Remedy: Reduced pressure (35 mbar) reduction of collision processes. Selection of a measuring range (650-850 nm) with low foreign absorption. Due to the high amplification factor of the photomultiplier (1:100.000) emission flanks can be used.