Acetic Acid Separation Methods
Acetic Acid Separation Methods. Supervisore : Prof . H. S. Ghaziaskar By: H. Rastegari. Contents. Acetic Acid Uses Acetic Acid Production Acetic Acid Separation methods Conclusion References. Acetic Acid Uses. In vinyl acetate monomer production In acetic anhydride production


Acetic Acid Separation Methods
E N D
Presentation Transcript
Acetic Acid Separation Methods Supervisore: Prof. H. S. Ghaziaskar By: H. Rastegari
Contents • Acetic Acid Uses • Acetic Acid Production • Acetic Acid Separation methods • Conclusion • References
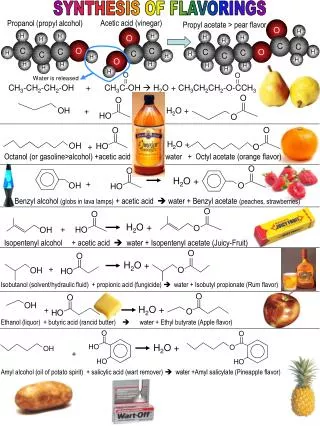
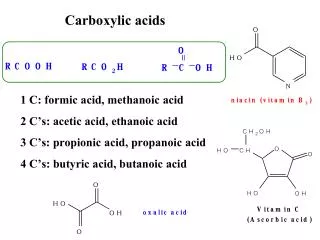
Acetic Acid Uses • In vinyl acetate monomer production • In acetic anhydride production • As solvent in production of terphetalic acid • As recrystalization solvent • In Silage • In production of various acetates such as: • Sodium acetate • Copper acetate • Aluminum acetate • Palladium acetate 2
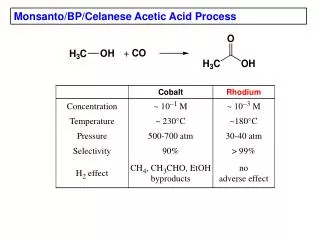
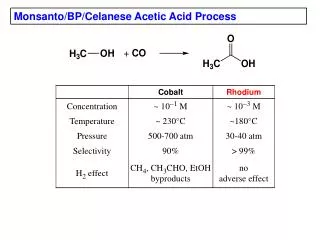
Acetic Acid Production • Chemical processes for acetic acid production: • Reaction of methanol with carbon monoxide • Reaction of acetylene with water followed by air oxidation • Fermentation of ethanol • Butane oxidation 3
Other chemical processes which produce acetic acid • as a by-product: • Manufacture of cellulose esters • Reactions involving acetic anhydride • Synthesis of glyoxal from acetaldehyde and nitric acid • Wood distillate 4
Problem • Separating acetic acid from water 5
Separation Methods • Separation Involving Phase Changes: • Simple Distillation • Azeotropic Distillation • Extractive Distillation • Reactive Distillation • Separation involving membranes: • Pervaporation • Evapomeation • Temperature Difference Evapomeation • Electrodialysis • Bipolar Membrane Electrodialysis
Simple Distillation • Physical separation process based on differences in volatilities 7
Advantage • Simple and easy to operate • Disadvantage • Large energy consumption 8
Azeotropic Distillation • Distillation in the presence of entrainer 9
Desirable properties for an azeotropic entrainer : • Heterogeneous azeotrope • Commercially available and inexpensive • Nontoxic • Chemically Stable • Noncorrosive • Low heat of vaporization The best entrainer is: Alkyl Acetate 10
Effective parameters for alkyl acetate selection • Azeotropic temperature • Azeotropic composition • Aqueous phase composition and entrainer pricing 11
Advantage Improving the economics of the separation • Disadvantage Requiring large amount of entrainer 12
Extractive Distillation • Distillation in the presence of solvent 13
Desirable properties for solvent: • Nonvolatile • High boiling point • Make large difference in volatility between components • Miscible with mixture and doesn´t form azeotropic mixture • Commercially available and inexpensive • Noncorrosive • Physically and chemically stable 14
The best solvent is: • Trialkyl Amine • Advantage Relatively little energy consumption • Disadvantage Need additional heat requirement on the column Some what larger plates 15
Reactive Distillation • Chemical separation method which combines simultaneous chemical reaction and multicomponent distillation in the same vessel 16
Mechanism of reaction: • First Step: Second Step: Third Step: 17
Effect of various parameters on the acid conversion • Total feed flow rate Optimum value:192 mL/h • Mole ratio 18
Reflux configuration • Feed position 19
Pervaporation (PV) • separation of liquid mixtures by partial vaporization through membrane 20
Used membrane • Polydimethylsiloxane (PDMS) • Cross-linked polybutadiene • Silicalite-1 as adsorbent filler in PDMS membrane • Carbon molecular sieve in PDMS membrane • Silicalite-1(pure silica) • Ge-ZSM-5 • Sn-ZSM-5 20
Effective parameters on separation performance • Si/Sn • Temperature • Acid concentration • Disadvantage Shrinking and swelling of the membrane 21
Evapomeation(EV) • Vaporization of feed solution then permeation through polymeric membrane 22
Effective parameters on separation performance • Temperature • Acid concentration • Disadvantage membrane condensation in high acid concentration 23
Temperature Difference Evapomeation (TDEV) • Decreasingtemperature in the membranesurroundingthaninthe feed solution 24
Electrodialysis (ED) • Ion transportation from one solution through ion- exchange membranes to another solution under the influence of an electric potential difference 25
Application Concentrating acetic acid from water containing %1(w/w) acid to %10(w/w) 26
Importance Make full use of our limited resources Protect our environment • DisadvantageConcentration efficiency up to %10 Low electric current efficiency (around %20) 27
Application Concentrating acetic acid from water containing %0.2(w/w) acid to %14(w/w) • Disadvantage Low electric current efficiency (around %40) 29
Conclusion For high purity (%99.9) acetic acidAzeotropic Distillation For reasonably pure acetic acid Extractive Distillation For ester production Reactive Distillation For separation from solution containing % (5-15) acid PV 30
References [1] Garwin,L., Hutchisoni,K., E., 1950. IndustrialAndEngineering Chemistry 42(4), 727-730. [2] Othmer, D., F., 1935. Industrial And Engineering Chemistry 27(3), 250-255. [3] Lee, F., M., Wytcherley, R., W., Distillation, Academic Press, USA, 2000. [4] Chien, I., L., Kuo, C., L., 2006. Chemical Engineering Science 61, 569-585. [5] Wang, S., J., Lee, C., J., Jang, S., S., Shieh, S., S., 2008. Process Control 18, 45-60.
[6] Garwin, L., Haddad, P., O., 1953. Industrial And Engineering Chemistry 45(7), 1558-1562. [7] Lei, Z., Li, C., Li, Y., Chen, B., 2004. Separation And Purification Technology 36, 131-138. [8] Taylor, R., Krishna, R., 2000. Chemical Engineering Science 55, 5183- 5229. [9] Saha, B., Chopade, S., P., Mahajani, S., M., 2000. Catalysis Today 60, 147-157. [10] Yu, L., Guo, Q., Hao, J., Jiang, W., 2000. Desalination 129, 283-288. [11] Sun, W., Wang, X., Yang, J., Lu, J., Han, H., Zhang, Y., Wang, J., 2009. Membrane Science 335, 83-88.
[12] Li, G., Kikuchi, E., Matsukata, M., 2003. Separation Purification Technology 32,199-206. [13] Toti, U., S., Kariduraganavar, M., Y., Soppimath, K., S., Aminabhavi, T., M., 2002. Applied Polymer Science 83, 259-272. [14] Asman, G., Anl, O., 2006. Separation Science And Technology 41(6) 1193-1209. [15] Asman, G., Sanli, O., 2006. Applied Polymer Science 100, 1385-1394. [16] Chien, I., L., Zeng, K., L., Chao, H., Y., Liu, J., H., 2004.Chemical Engineering Science 59, 4547-4567. [17] Kittur, A., A., Tambe, S., M., Kulkarni, S., S., Kariduraganavar, M., Y., 2004. Applied Polymer Science 94, 2101-2109.
Acetic Acid Production Glyoxal:
Simple Distillation VHAC= (yHAC) / (xHAC) VH2O= (yH2O) / (xH2O) Dalton's Law: PH2O= (yH2O) * pt Raoult's law: PH2O= (xH2O) * p0H2O
α = VH2O / VHAC Fenske Equation: (yH2O)/(1- yH2O) = αn+1 (xH2O)/(1- xH2O)
Extractive Distillation Solvent volume