Insulin Pathway
Insulin Pathway. SIGMA-ALDRICH. Insulin Pathway

Insulin Pathway
E N D
Presentation Transcript
Insulin Pathway SIGMA-ALDRICH
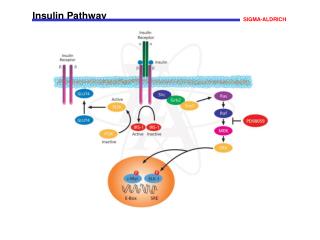
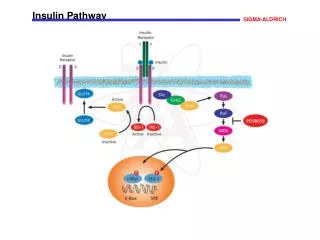
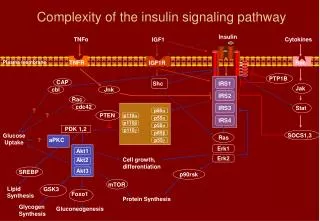
Insulin Pathway Signaling through the insulin pathway is critical for the regulation of intracellular and blood glucose levels and the avoidance of diabetes. Insulin binds to its receptor leading to the autophosphorylation of the ß-subunits and the tyrosine phosphorylation of insulin receptor substrates (IRS). IRS phosphorylates the SH2 domain of Shp2, a tyrosine phosphatase, and the SH3 domain of the adaptor molecule Grb2. Activated Grb2 recruits Sos1 that, in turn, activates the Ras signaling pathway and gene transcription. IRS also activates phosphoinositide 3-kinase (PI3K) through its SH2 domain, thus increasing the intracellular concentration of PIP2 and PIP. This, in turn, activates phosphatidylinositol phosphate-dependent kinase-1 (PDK-1), that subsequently activates Akt/PKB This results in the translocation of the glucose transporter (GLUT4) from cytoplasmic vesicles to the cell membrane. References Bevan, P., Insulin signaling. J. Cell Sci., 114, 1429-1430 (2001). Kido, Y., et al., Clinical review 125: The insulin receptor and its cellular targets. J. Clin. Endocrinol. Metab., 86, 972-979 (2001).