Electric Charge
Electric Charge. Chapter 17.1. Examples of Electric Charge. Rubbing a plastic comb through your hair. Rubbing a balloon on your hairs. Rubbing your shoes across a wool rug. Sliding your clothing across a car seat.


Electric Charge
E N D
Presentation Transcript
Electric Charge Chapter 17.1
Examples of Electric Charge • Rubbing a plastic comb through your hair. • Rubbing a balloon on your hairs. • Rubbing your shoes across a wool rug. • Sliding your clothing across a car seat. • Experiments like these work best when the air is dry, moisture creates a pathway for the charge to leak off the materials.
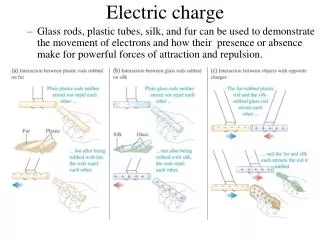
Two Kinds of Charge • There is a Positive Charge (+) and a negative charge (-). • Benjamin Franklin (1706-1790) coined the term positive and negative. • Opposites attract and likes repel.
Positive & Negative • When rubbing a balloon across your hair, the balloon becomes negatively charged and the hair becomes positively charged. • Positive and negative are said to be opposite because an object with an equal amount of positive and negative charge has no net charge.
Atoms • An atom is composed of three parts: • Protons: Positively Charged (+) • Neutrons: Neutral Charge • Electrons: Negatively Charged (-) • The protons and neutrons are relativity fixed in the atom, but the electrons located on the outside of the atom can be transferred from one atom to another quite easily.
Charged Ions • When electrons are balanced with the protons, the atom has no net charge. • If an electron is transferred from one neutral atom to another, one of the atoms gains a negative charge and the other loses a negative charge, therefor becoming positive. • Ions – Atoms with a net positive or negative charge.
Balloon and Hair • Rub the balloon against hair and a charge starts to build. • The balloon pulls electrons away from the hair • Hair loses electrons and becomes positively charged. • Balloon gains electrons and becomes negatively charged. • By increasing the surface area of the objects, the charges can be increased. • The net positive charge will be equal to the net negative charge.
Millikan Oil Drop Experiment • Robert Millikan (1886-1953) performed an experiment by levitating a charged oil droplet in an electric field. • From the experiment he learned that electric charge is quantized. • Multiples of a fundamental unit of charge. • Electrons have a charge of –e • Protons have a charge of +e
Charges • The value of electric charge: e = 1.60219x10-19 C • The SI Unit for charge is a Coulomb (C) -1C = 6.2x1018 Electrons
Transfer of Electric Charge • Depending on the ability for electrons to flow in a material determines if the material is a conductor or an insulator. • A rubber balloon can receive a net charge, but the charge can not flow freely around the balloon, therefore it’s an insulator. • Copper can receive a net charge, but the charge can flow through the material making it a good conductor.
Conductors • Conductor – Material that transfers charge easily. • Outer electrons of the atoms in a metal are not anchored to the nuclei of particular atoms, but are free to roam in the material. • Metals are good conductors for the motion of electric charges because their electrons are “loose.” • Ex: • Copper • Gold • Aluminum • Silver
Insulators • Insulator – Material that does not transfer charge easily. • Electrons insulators are tightly bound and remain with their particular atoms. • They are not free to wander about to other atoms in the material. • Ex: • Rubber • Silk • Glass • plastic
Semiconductors & Superconductors • Semiconductors are a material characterized by electrical properties between insulators and conductors. • Atoms in a semiconductor hold their electrons until given small energy boosts. • This occurs in photovoltaic cells that convert solar energy into electrical energy. • Germanium and silicon • Superconductors are materials that have no resistance to the follow of electrons. • Super cooling metals
Charge by Contact • Electrons can also be transferred from one material to another by simply touching. • When a charged rod is placed in contact with a neutral object, some charge will transfer to the neutral object. • This method of charging is called charging by contact. • If the object is a good conductor, the charge will spread to all parts of its surface because the like charges repel each other.
Induction • Induction – The process of charging a conductor by bringing it near another charged object and grounding the conductor.