Empirical Formula vs. Molecular Formula
60 likes | 501 Vues
Empirical Formula vs. Molecular Formula. Empirical formula: the formula for a compound with the smallest whole-number mole ratio of the elements

Empirical Formula vs. Molecular Formula
E N D
Presentation Transcript
Empirical Formula vs. Molecular Formula • Empirical formula: the formula for a compound with the smallest whole-number mole ratio of the elements • Molecular formula: formula for a compound with the actual number of atoms of each element in a molecule or formula unit of the substance • What is the difference between a molecule and a formula unit?
Empirical Formula vs. Molecular Formula, continued…… • Example of the difference: Benzene is composed of 6 carbon and 6 hydrogen (C6H6). • What is the ratio of carbon-to-hydrogen in benzene? • If we reduce the compound formula to its “smallest whole-number mole ratio of the elements” (empirical formula), we are left with CH • Acetylene’s molecular formula is C2H2. What is its empirical formula?
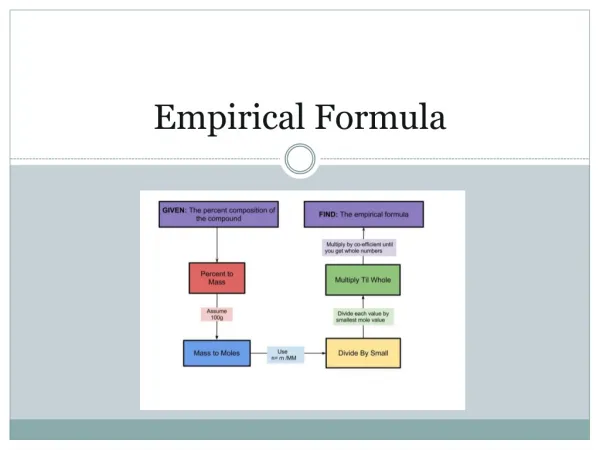
Calculating Empirical Formula From Percent Composition • If given percent composition of a compound, make the percent of each element that element’s mass in grams, so the whole compound has a molar mass of 100 grams. • Convert the mass of each element to moles. • If the answers (moles) are not whole numbers, divide all answers by the smallest number of moles present. • If still not whole numbers, multiply all answers by the smallest factor that will give you whole numbers.
Calculating Empirical Formula From Percent Composition, continued….. • Examples: P.349 (144, 147) • Practice Problems (p.333) --- reference Example Problem on p.332 for needed guidance