Understanding Empirical and Molecular Formulas: Calculation Methods and Examples
This guide explains empirical formulas (EF) and molecular formulas (MF), highlighting how to determine the simplest atom ratios from percentage composition or mass. Through examples including hydrogen peroxide (H2O2) and others, the process involves calculating moles from mass, dividing by the smallest number, and formulating the simplest whole number ratios. Additionally, it covers converting EF to MF by calculating molar masses and using ratios. Practice problems and their solutions support understanding of these fundamental chemistry concepts.


Understanding Empirical and Molecular Formulas: Calculation Methods and Examples
E N D
Presentation Transcript
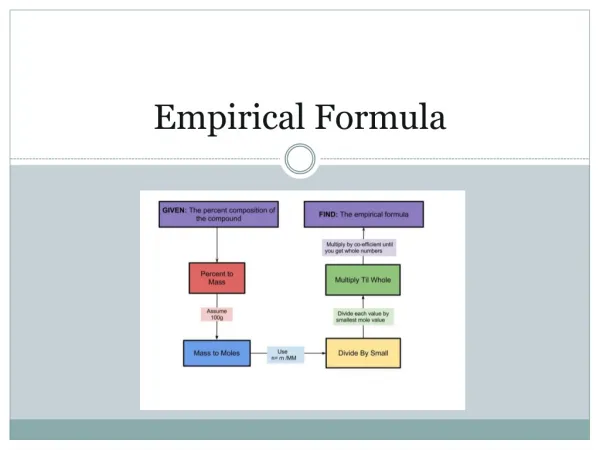
Empirical Formula • The simplest ratio of atoms • For example, the empirical formula of H2O2 (hydrogen peroxide) is HO (1 hydrogen atom for every oxygen atom)
How to calculate EF • From percentage composition or mass E.g. 49.5g C, 5.2g H, 28.8g N, 16.5g O • Arrange the elements in % or mass Done • Divide each by the molar mass of the element to get moles C49.5 H5.2 N28.8 O16.5 12 1 14 16
C 4.121 H 5.159 N 2.056 O 1.031 • Divide everything by the SMALLEST NUMBER 4.121 5.159 2.056 1.031 1.031 1.031 1.031 1.031 = 4 : 5 : 2 : 1 • Write the formula with the simplest whole number ratio C4H5N2O ANSWER
Now try these! • Nutrasweet is 57.14% C, 6.16% H, 9.52% N and 27.18% O. • A compound is 72.2% Mg and 27.8% N • An oxide of nitrogen contains 42.05 g N and 95.95 g of O. • Mercury forms a compound that is 73.9% Hg and 26.1% Cl. • Vitamin C contains 40.92% C, 4.58% H and 54.50% O.
Now try these - ANSWERS • C14H18N2O5 • Mg3N2 • NO2 • HgCl2 • C3H4O3
From EF to molecular formula • The molecular formula is the ACTUAL NUMBER OF EACH ATOM in the compound
How to work out the MF • Calculate the molar mass for the empirical formula. Example: for a compound where there EF is HO and actual molar mass is 34 gmol-1 Molar mass of EF = 1 + 16 = 17 gmol-1 • Divide the molar mass MF ÷ EF Actual MF ÷ EF 34 ÷ 17 gmol-1 = 2 • Multiply the formula by the factor calculated in part 2. HO x 2 = H2O2
Questions – Molecular formula • A compound has an empirical formula of CH2Cl and a molar mass of 99 gmol-1. Calculate the molecular formula. • A compound has 75.46% C, 4.43% H and 20.1% O and a molar mass of 318 gmol-1. Calculate the molecular formula.
Answers – Molecular formula • The EF is CH2Cl. The molar mass for this is 12 + 2 x 1 + 35.5 = 49.5. 99 ÷ 49.5 = 2, therefore the molecular formula is TWICE that of the EF – C2H4Cl2. • The EF is C10H7O2. The molar mass for this is 12 x 10 + 7 x 1 + 2 x 16 = 159. 318 ÷ 159 = 2, therefore the molecular formula is TWICE that of the EF – C20H14O4*.